This patent landscape report explores global innovation trends in CRISPR (Clustered Regularly Interspaced Short Palindromic Repeats) genome editing technologies, with a specific focus on their application in human therapeutics. CRISPR has revolutionized genetic engineering by enabling precise, efficient, and cost-effective gene modifications. Originally derived from bacterial immune systems, CRISPR—especially the Cas9 variant—has seen widespread adoption in medical, agricultural, and pharmaceutical research.
Focusing on the period from 2014 to 2024, this report examines the exponential growth in patent filings and grants, emphasizing developments in human genome editing, both somatic and germline. It assesses the role of CRISPR in addressing genetic disorders such as cancer, sickle cell disease, and congenital blindness. The analysis includes key metrics such as major assignees, patent jurisdictions, technological domains, and industry sectors.
Beyond quantitative trends, the report delves into qualitative factors shaping the innovation landscape: evolving CRISPR tools like Cas12, Cas13, base editing, and prime editing; major legal battles over intellectual property rights; and the emergence of personalized gene therapies. These insights not only reveal competitive dynamics but also underscore the ethical, regulatory, and societal implications of CRISPR-based innovation. Through this comprehensive analysis, the report aims to map the trajectory of CRISPR technologies and assess their transformative potential in biotechnology and public health.
Objectives
- Examine the global patent filing trends related to CRISPR genome editing in humans from 2014 to 2024.
- Identify key assignees, assignee countries, and priority areas in the CRISPR patent landscape.
- Analyse the main International Patent Classification (IPC) codes associated with CRISPR technologies.
- Assess the distribution of patents across major industries and technological domains.
- To understand the advancements in CRISPR tools (Cas9, Cas12, Cas13, base editing, prime editing) and their impact on innovation.
- Evaluate real-world applications and ethical considerations through notable case studies like Baby KJ.
- Explore the implications of patent disputes and licensing on future CRISPR-based therapies and global research efforts.
What is Human Genome Editing?
Human Genome Editing refers to changing DNA in a specific way. It can add, remove, or fix DNA in the genome. It can be applied in 3 ways – somatic cells (not passed to children),germline cells(not for reproduction), germline cells (for reproduction-heritable).
 The application of somatic human genome editing has been undertaken, including in vivo editing ,to address HIV and sickle-cell disease , for example. Even though somatic human genome editing is accepted in many countries, and they are many benefits, challenges still preside. One major challenge is creating innovations that consider the diversity of the human population and experience.[1] Other challenges that preside are medical travel , unethical clinics and the reporting of illegal, unregistered, unethical, or unsafe research.
The application of somatic human genome editing has been undertaken, including in vivo editing ,to address HIV and sickle-cell disease , for example. Even though somatic human genome editing is accepted in many countries, and they are many benefits, challenges still preside. One major challenge is creating innovations that consider the diversity of the human population and experience.[1] Other challenges that preside are medical travel , unethical clinics and the reporting of illegal, unregistered, unethical, or unsafe research.
Whereas heritable human genome editing is referred when nuclear DNA is edited in a way that is heritable across generations. However, this type of editing has be severely debated over in regard to the possible consequences for offspring and for society in general. It may also cause safety and ethical issues.
Therefore, somatic human genome editing is mostly preferred when compared to heritable human genome editing.[2]
What is CRISPRR Genome Editing ?
The National Human Genome Research Institute states CRISPR (clustered regularly interspaced short palindromic repeats) to be “ a technology that aids researchers in the selective modification of the DNA of living organisms. CRISPR was adapted for laboratory use from naturally occurring genome editing systems found in bacteria.”[3]
CRISPR has been used by scientist to add, remove or change parts of DNA to fix problems and understand diseases presiding in the DNA.
Currently clinical test is being undertaken revolving CRISPR especially regarding cystic fibrosis, sickle cell disease, cancer, and HIV. There are hopes that CRISPR will help aid and provide treatment for these major illnesses.
A natural defence system in bacteria has also led to another major invention CRISPR-Cas9. It is currently used as a special guide for RNA so that it can find the exact spot in the DNA to cut. The Cas9 enzyme is then used to cut the DNA and make the required modifications.[4]
There have also been some ethical concerns such as if the modified DNA would be continued by future changes and the effects presiding in embryos, sperm, and egg cells. Due to these challenges some countries have been reluctant to allow human genome editing in their country.[5]
What is CRISPR -Cas9 ,Cas12,Cas13?
CRISPR-Cas9 can be considered as a pair of scissors it cuts DNA and makes the required modifications. It identifies a particular disease and either adds, changes or removes something in the DNA to solve the problem. The changes made by this type of technology remain permanent and preside in the body forever. However, it should be noted that the technology can sometimes also cut the wrong part of the DNA which can cause major problems.
CRISPR -Cas12 can also be used to modify DNA, however it makes staggered cuts instead of straight cuts like CRISPR- Cas9. It can also cut single-stranded DNA nearby. This also aids in finding major viruses and diseases. It is also considered to be much easier to use than the other types of CRISPR technologies. It is useful in research and testing as it can modify many genes at once.
CRISPR-Cas13 is different from the other two types of technologies as it makes changes to RNA instead of DNA. It prevents the creation of disease making proteins. Therefore, the changes are only considered temporary as changes are to RNA instead of DNA.
In summary, Cas9 and Cas12 are used for permanent changes in DNA, while Cas13 is used for temporary changes in RNA. Scientists choose between them based on whether they want to change DNA forever or just temporarily stop harmful effects.
Parameters used for Patent Landscape analysis.
- Patent filing trend between years 2014 to 2024
- Distribution of Patents based on the Assignees.
- Jurisdiction based Global Patent Filing Trends
- Technology classification based on IPC classification.
- Activities of Competitors based on area of Research (IPC)
- Estimated top ten industries and technology areas between 2014 and 2024.
- Estimated top 10 owners between 2014 and 2024.
- Estimated top 10 Assignee Countries between 2014 and 2024
- Estimated top 10 Priority Countries between 2014 and 2024
- Estimated top 10 Publication Countries between 2014 and 2024
Patent filing trend between years 2014 to 2024
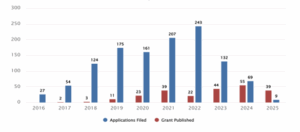 Figure 1 : Patent filing trend between the years 2014 to 2024.
Figure 1 : Patent filing trend between the years 2014 to 2024.
Figure 1 illustrates the trend in patent activity related to CRISPR human genome editing from the year 2014 to 2024, covering both applications filed and grants published. A steady increase in patent filings is observed from 2016, peaking in 2022 with 243 applications, followed by a decline in 2023 (132) and 2024 (69). The number of patents granted remained low in the early years, with only 2 to 3 grants annually between 2016 and 2018. However, a notable rise occurred from 2020 onwards, with the highest number of grants (44) published in 2023, followed by 39 in 2021 and 22 in 2022. While 2022 saw the most applications filed, the highest number of grants was recorded in 2023, indicating a time lag between filing and grant. Overall, the data suggests a significant surge in innovation and interest in CRISPR genome editing technologies, particularly between 2018 and 2022, followed by a slight decline post-2022.
Distribution of Patents based on the Assignees.
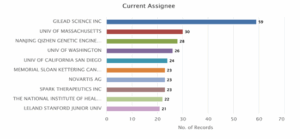
Figure 2 presents the distribution of current assignees for patents related to CRISPR human genome editing. Gilead Sciences Inc. leads significantly with 59 patent records, indicating a dominant role in the field. This is followed by the University of Massachusetts with 30 patents and Nanjing Qizhen Genetic Engineering Co. Ltd. with 28 records. Other notable contributors include the University of Washington (26) and University of California San Diego (24). Several institutions such as Memorial Sloan Kettering Cancer Centre, Novartis AG, and Spark Therapeutics Inc. each hold 23 patents, highlighting strong industry and academic involvement. The National Institutes of Health (22) and Stanford University (21) also feature among the top assignees. Overall, the chart highlights a balanced mix of academic, public, and private sector innovation in advancing CRISPR genome editing technologies.
Jurisdiction based Global Patent Filing Trends
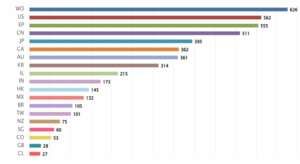
Figure 3: Distribution of Patent based on the jurisdiction.
Figure 3 shows the distribution of patents based on the jurisdiction in which they were filed. The highest number of patents were filed under the World Intellectual Property Organization (WO) with 626 applications, followed by the United States (US) with 562 patents and the European Patent Office (EP) with 555 patents. China (CN) and Japan (JP) also contributed significantly, with 511 and 395 patents, respectively. Other notable jurisdictions include Canada (CA) with 362, Australia (AU) with 361, and South Korea (KR) with 314 patents.
Technology classification based on IPC classification.
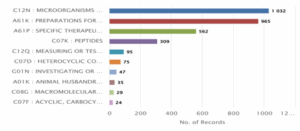
Figure 4: CRISPR Genome Editing in Humans Patent Distribution based on main IPC Code Classification
Figure 4 displays the distribution of patents across different International Patent Classification (IPC) codes relevant to CRISPR human genome editing. The highest number of patents fall under C12N: Microorganisms or Enzymes, with a total of 1,032 records, indicating a strong focus on genetic manipulation and biotechnology. This is closely followed by A61K: Preparations for Medical, Dental or Toilet Purposes with 965 records, and A61P: Specific Therapeutic Activity of Chemical Compounds or Medicinal Preparations with 562 records, both highlighting the medical and therapeutic applications of CRISPR. Other significant IPC codes include C07K: Peptides (309) and C12Q: Measuring or Testing Processes Involving Enzymes or Microorganisms (95). Categories like C07D: Heterocyclic Compounds (75) and G01N: Investigating or Analysing Materials (47) also appear prominently. Lesser but notable classifications include A01K: Animal Husbandry (35),
C08G: Macromolecular Compounds (29), and C07F: Acyclic, Carbocyclic Compounds (24). This data indicates a diverse yet concentrated research focus, primarily in medical, biochemical, and microbiological domains.
Activities of Competitors based on area of Research (IPC)
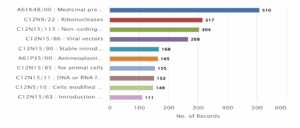
Figure 5: CRISPR Genome Editing in Humans – Patent based on Competitors and Area of Research (IPC)
Figure 5 illustrates the IPC Full-level classification of patents related to CRISPR human genome editing, highlighting the specific technologies being targeted. The leading IPC category is A61K48/00: Medicinal preparations containing genetic material with 510 records, reflecting the dominant focus on gene-based therapies. This is followed by C12N9/22: Ribonucleases (317) and C12N15/113: Non-coding nucleic acids (304), indicating extensive research into RNA-related technologies and genetic regulation mechanisms. Other significant areas include C12N15/86: Viral vectors (268) used for gene delivery, and C12N15/90: Stable introduction of genetic material into eukaryotic cells (168). Additionally, A61P35/00: Antineoplastic agents (165) show the therapeutic targeting of cancer using CRISPR. Categories such as C12N15/85 (for animal cells – 155), C12N15/11 (DNA or RNA fragments – 152), and C12N5/10 (modified animal cells – 149) further demonstrate the cellular and molecular focus of these innovations. Finally, C12N15/63: Introduction of foreign genetic material using vectors with 111 patents emphasizes the technical approach in CRISPR delivery mechanisms. Overall, the graph confirms that gene therapy and genome editing applications form the core of CRISPR-related research.
Estimated top 10 industries and technology areas between 2014 and 2024.
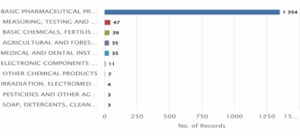
Figure 6: CRISPR Genome Editing in Humans – Distribution of Patent based on Industries & Technology.
Figure 6 illustrates the distribution of patents across major industries and technology areas from 2014 to 2024 in the domain of CRISPR human genome editing. The dominant industry by a large margin is Basic Pharmaceutical Products and Preparations, with 1,354 patent records, indicating the primary application of CRISPR in drug development and therapeutics. This is followed by Measuring, Testing and Navigating Equipment with 47 records, and Basic Chemicals, Fertilisers, and Nitrogen Compounds with 39 records. Other notable sectors include Agricultural and Forestry Machinery (35), Medical and Dental Instruments (35), and Electronic Components and Boards (11). Smaller contributions come from industries such as Other Chemical Products (7), Irradiation and Electromedical Equipment (4), and Pesticides and Other Agrochemical Products (3). The Soap, Detergents, and Cleaning Products industry recorded the least number of patents, with just 3 records. Overall, the graph emphasizes the biopharmaceutical and healthcare sector’s dominance in CRISPR-related innovations, with limited but emerging applications across agriculture, electronics, and chemical industries.
Estimated top 10 owners between 2014 and 2024.
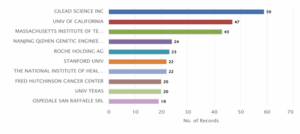
Figure 7: CRISPR Genome Editing in Humans – Distribution of Patent based on Current Owner
Figure 7 highlights the key assignees based on the number of patent records. Gilead Science Inc. emerges as the leading assignee with 59 patents, followed by the University of California with 47 and the Massachusetts Institute of Technology with 43.
Other significant contributors include Nanjing Qizhen Genetic Engineering Co. Ltd. with 24 patents, Roche Holding AG with 23, and both Stanford University and The National Institute of Health with 22 each.
Additionally, Fred Hutchinson Cancer Centre and University of Texas have each filed 20 patents, while Ospedale San Raffaele SRL rounds out the top assignees with 19 records.
Estimated top 10 Assignee Countries between 2014 and 2024
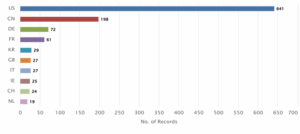
Figure 8: Distribution of Patent based on Assignee Countries
Figure 8 displays the distribution of patents based on the assignee countries. The United States (US) holds the highest number of records with 641 patents, making it the leading jurisdiction. It is followed by China (CN) with 198 patents and Germany (DE) with 72.
Other significant contributors include France (FR) with 61 patents, South Korea (KR) with 29, and both the United Kingdom (GB) and Italy (IT) with 27 each. Ireland (IE) and Switzerland (CH) recorded 25 and 24 patents respectively, while Netherlands (NL) accounted for 19.
Estimated top 10 Priority Countries between 2014 and 2024
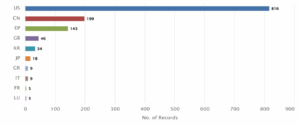
Figure 9: Distribution of Patent based on Priority Countries
Figure 9 shows the distribution of patents based on the priority country. The United States (US) holds the largest number of priority filings, with 816 patent records, followed by China (CN) with 199 and the European Patent Office (EP) with 143.
Other key priority jurisdictions include the United Kingdom (GB) with 46, South Korea (KR) with 34, and Japan (JP) with 18. Meanwhile, Greece (GR) and Italy (IT) contributed 9 patents each, while France (FR) and Luxembourg (LU) had 5 each, representing the lower end of the distribution.
Estimated top 10 Publication Countries between 2014 and 2024
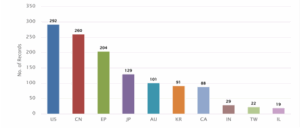
Figure 10 : Distribution of Patent based on Published Countries
Figure 10 represents the distribution of patents based on the inventor’s country. The United States (US) accounts for the highest number of records, with 292 patents, followed closely by China (CN) with 260 and the European Patent Office (EP) with 203.
Other significant contributors include Japan (JP) with 128 patents, Australia (AU) with 100, South Korea (KR) with 91, and Canada (CA) with 88 records.
Further, India (IN) accounts for 29 patents, followed by Taiwan (TW) with 22 and Israel (IL) with 19, representing the lower range of inventor country contributions.
CRISPR and Human Health
CRISPR aims to eliminate diseases caused by human genes it does this through many means :
- Blood Disorders– Scientist identify the faulty cells and take them to the lab for modification. After the needed modifications are made, they are returned into the patient’s body. Results have proved to be effective.
- Blindness – In 2020 ophthalmologists used CRISPR for the first time to cure blindness in their patients. They inject CRISPR directly in the eye to find the rare blindness causing gene and remove it. Ophthalmologists say if this proves to be a safe means they will continue using it.
- Cancer – Doctors remove the cancer-causing cells , CRISPR is then used to make the cells have a better chance at fighting the cancer. The cells are then kept back in the body. It was found that the cancer didn’t grow as much, and the treatment was safe.
- Fast Diagnosis – CRISPR is used to find viruses and diseases in a very quick manner. It helped create the first FDA-approved COVID-19 test using CRISPR.
CRISPR is still being tested, but it offers hope for treating many diseases—from rare genetic conditions to common illnesses like cancer and viral infections.[7]
CRISPR Modifications and Advancements
CRISPR technology has evolved over time with the system being more accurate, faster, and efficient in detecting human genome diseases. The major upgrades are :
- Modified Cas9 Proteins – Cas9 is used to add , remove or change DNA and detect diseases. Recently, scientist have made new inventions such as nCas9 (nickase Cas9) which only cuts one strand of DNA. dCas9 (dead Cas9) instead of cutting DNA it binds it. These two strategies have been proven to be a safer means of human genome editing. This can be attached to editing enzymes to change DNA without breaking it.
- Base Editing – This uses nCas9 or dCas9 + adeaminase enzyme and can change one letter in DNA for example “A to G” etc. This method has proved to be effective as there is no double- strand breaks. However, one limitation is that not all DNA can be edited.
- Prime Editing – This has proven to be a newer and more effective means to human genome editing. It uses nCas9+ reverse transcriptase+ special guide RNA (pegRNA).It is known to correct more types of mutations and correct 89% of human genetic mutations.
- RNA Editing – It uses Cas13b+ adenosine deaminase. It edits RNA and therefore is much safer because the edits are not permanent. It is useful for diseases where temporary fixes are enough.
- Easier Delivery Systems –To treat people as these tools enter the cells and are often used to detect major diseases like AAV The initial problem was that Cas9 is too big for AAV. The solution was a smaller enzyme called AsCas12f which is 1/3 of Cas9. This proved fit inside AAV and was proved to successfully when assessed on mice.
- Why This Matters for Human Health
- Fewer mistakes when editing genes.
- More types of genetic diseases can be treated.[8]
- Better delivery into the human body.
- Safe and specific therapies can be developed for individual patients.
CRISPR Patent Dispute
The discovery of CRISPR- Cas9 has led to a major patent battle between 2 parties. One of the major reasons for the dispute was financial stakes and who would have authority over the technology especially regarding human cells.
The two parties were Jenifer Doudna from the University of California, Berkely and Emmanuelle Charpentier from the University of Vienna & Umeå University who filed their patent in May of 2012.
The other party was Feng Zhang from the Broad Institute of MIT and Harvard. He filed his patent in December of 2012,just as his paper showing CRISPR use in human cells was accepted.
By April 2014 Zheng’s team was granted the first patent while Doudna’s was still under review. This led to Doudna’s team challenging the decision.
Eventually by 2019 both parties were granted patent access, biotech companies would either get licenses from Doudna or Zhang.
However, in February of 2022 the U.S Patent Office sided with Zhang. This cause major financial problems for Doudna’s team.
It should be note that in Europe and other countries Doudna and Charpentier won and hold major patents.
- Why Does It Matter?
- The patent controls who can profit from using CRISPR for human gene editing.
- It affects:
-
- Biotech companies creating CRISPR-based therapies.
- Patients waiting for treatments.
- Global research and licensing costs.[9]
Current News – The case of Baby KJ
The Children’s Hospital of Philadelphia and Penn Medicine hit a major milestone in February 2025 by using CRISPR to treat a baby named KJ. KJ was born with a rare genetic condition called CPS1 deficiency. The condition stops the body from breaking down harmful ammonia and if left untreated it cause major brain and liver damage.[10]
Due to KJ’s young age a liver transplant was not feasible, doctors created a personalised CRISPR therapy accustomed to him .
They used base editing for the process to correct the faulty gene present in his liver. his therapy was delivered through lipid nanoparticles and given in three doses by April 2025.
KJ had responded well:
- He could eat more protein without harmful effects.
- He needed fewer medications.
- He recovered from childhood infections without serious complications.
- No serious side effects had occurred.
The treatment marked the first CRISPR had been personalised, it showed CRISPR could go beyond common diseases and could tackle unique or ultra-rare mutations.
Doctors hope that this success can pave the way for more personalised CRISPR treatments for many diseases that currently have no cure.
However, long-term follow-up is still needed to see how well it works overtime.
Analysis of Report
The patent landscape analysis clearly shows a significant surge in CRISPR-related innovation between 2014 and 2024, particularly from 2018 to 2022. The peak in applications in 2022 and the rise in grants by 2023 indicates both growing research interest and maturing technologies ready for real-world use.
Key players such as Gilead Sciences Inc., the University of California, and the Massachusetts Institute of Technology dominate the patent filings, suggesting strong competition between academic institutions and private companies. The data also shows that countries like the United States, China, Japan, and Germany are leading both in terms of assignee location and inventor nationality, underlining their role as global innovation hubs in biotechnology.
From an industrial perspective, Basic Pharmaceutical Products and Preparations hold the largest number of patents, confirming that CRISPR is primarily being applied in drug development and treatment of genetic diseases. The IPC classification further reflects this, with a major focus on gene therapy, viral vectors, and non-coding nucleic acids.
The analysis also highlights technological diversification with the evolution of CRISPR tools like Cas9, Cas12, Cas13, base editing, and prime editing, all of which aim to increase precision and safety. These advancements are essential in addressing ethical and clinical concerns, particularly in sensitive areas like embryo editing.
Moreover, real-world applications such as the case of Baby KJ demonstrate how CRISPR is transitioning from experimental use to personalised medicine, offering hope for rare and otherwise untreatable conditions.
However, the report also points to ethical debates, regulatory inconsistencies, and intellectual property disputes that may shape or limit access to CRISPR-based treatments. The patent conflict between Doudna and Zhang reflects the high commercial stakes and the complexity of navigating ownership in a rapidly evolving field.
Conclusion
CRISPR genome editing has shown rapid growth between 2014 and 2024, with a strong focus on pharmaceutical and medical applications. The rise in patent filings and technological advancements like Cas9, Cas12, and prime editing reflect its potential in treating genetic disorders.
While countries like the US and China lead in innovation, ethical concerns, patent disputes, and global regulations remain key challenges. Real-world cases, such as Baby KJ, highlight the shift towards personalised CRISPR therapies, offering hope for rare and serious conditions.
Overall, CRISPR stands as a powerful tool in modern medicine, but its future depends on responsible innovation and global cooperation.
Disclaimer
This landscape report is based on resources available in public domain such as published patent applications and granted patents freely available/accessible/downloadable generally paid and Government based patent databases unless specified otherwise. Moreover, the accuracy of the records kept and maintained by the respective patent offices and registries. As such, we are not responsible for any error, omission, and mistake or misfiled particulars contained or omitted from the said records. It must also be noted that the said records may not be updated on a regular basis.
Furthermore, the report is based upon individual expert’s view/judgment. Such analysis may vary from expert to expert. Experts’ views may not concur with/should not be treated as the views of the Author.
Author: Ishika Sarah Koshy , REVA University, Bangalore
Please contact us at info@origiin.com to know more about our services (Patent, Trademark, Copyright, Contract, IP Licensing, M&A of companies)
Subscribe to YouTube Channel HERE
Join LinkedIn Group: Innovation & IPR
WhatsApp: +91 74838 06607



