Jul 19, 2025 | Patent
 The growing global demand for clean and sustainable energy has driven significant interest in harnessing the power of the oceans. Sea energy, encompassing technologies such as wave energy, tidal energy, ocean thermal energy conversion (OTEC), and salinity gradient power, represents a promising frontier in renewable energy innovation. As countries and corporations invest in marine energy solutions, the patent landscape has become a critical indicator of technological advancements, competitive positioning, and commercialization potential.
The growing global demand for clean and sustainable energy has driven significant interest in harnessing the power of the oceans. Sea energy, encompassing technologies such as wave energy, tidal energy, ocean thermal energy conversion (OTEC), and salinity gradient power, represents a promising frontier in renewable energy innovation. As countries and corporations invest in marine energy solutions, the patent landscape has become a critical indicator of technological advancements, competitive positioning, and commercialization potential.
Key Technologies of Sea Energy
- Wave Energy Conversion (WEC) captures energy from ocean surface waves using devices like oscillating water columns, point absorbers, and attenuators. This technology dominates the marine energy patents and focuses on floating vs. fixed devices, power take-off (PTO) mechanisms, and wave farm optimization.
- Tidal energy extracts energy from tidal currents using underwater turbines (similar to wind turbines) or tidal barrages and uses this in innovations such as blade design, mooring systems, and subsea infrastructure.
- Ocean thermal energy conversion (OTEC) uses temperature differences between warm surface water and cold deep water to generate electricity. This technology has lower patent activity compared to wave/tidal but is growing in tropical regions.
- Salinity gradient power (Blue Energy) generates power from the difference in salt concentration between seawater and freshwater (e.g., using reverse electrodialysis or pressure-retarded osmosis). This is an emerging technology with niche patent filings.
- Offshore floating solar (Hybrid Systems) combines solar PV with floating platforms in calm sea regions. This technology overlaps with wave energy patents.
The report delves into the patent landscape of Sea energy, providing key insights into key players, technological trends and the competitive dynamics shaping the future of sea energy. By analyzing patent filings and grant trends, it aims to offer a comprehensive view of the intellectual property landscape that is driving the Sea energy forward.
OBJECTIVE OF PATENT LANDSCAPE
The objective of this patent landscape report for sea energy is to understand various trends with regard to patent activity in this space. The reports analyzed the patents filed, published and granted between 2014 and 2024. The search strategy used to generate data to prepare the report is based on a combination of predetermined International Patent Classification (IPC) codes, as well as specific keywords.
PARAMETERS USED FOR PATENT LANDSCAPE ANALYSIS
In this report, we studied the following parameters and trends:
- Patenting Trends for the year 2016 to 2024
- Distribution of Patents based on the Assignees
- Jurisdiction based Global Patent Filing Trends
- Technology classification based on IPC classification
- Activities of Competitors based on area of Research (IPC)
- Estimated top 10 industries and technology areas between 2014 and 2024
- Estimated top 10 owners between 2014 and 2024
- Estimated top 10 Assignee Countries between 2014 and 2024
- Estimated top 10 priority countries between 2014 and 2024
- Estimated top 10 publication countries between 2014 and 2024
A. PATENTING TRENDS BETWEEN THE YEAR 2014 TO 2024
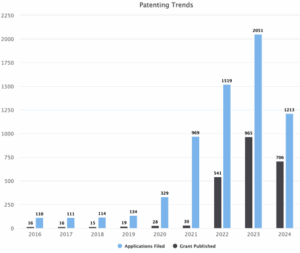
Figure 1: Sea Energy Patent filing trends between the years 2016 to 2024
Figure 1 depicts the distribution of patents for Sea energy between the years 2016 to 2024, including both applications filed and granted patents. The graph shows that the highest number of applications were filed in 2023 with 2051 patents followed by 2022 with 1519 patents and 2024 with 1213 patents. In the rest of the years i.e., 2021 is 969, 2020 is 329, 2019 is 134, 2018 is 114, 2017 is 111 and 2016 is 110. The highest number of patents granted in 2023 with 965 patents followed by 2024 with 706 patents and 2022 with 541. In the rest of the years i.e., 2021 is 30, 2020 is 28, 2019 is 19, 2018 is 15, 2017 is 16 and 2016 is 16. Overall, fillings for Sea energy patent families is gradually increased in the time period between 2016 and 2024.
B. DISTRIBUTION OF PATENTS BASED ON THE ASSIGNEES
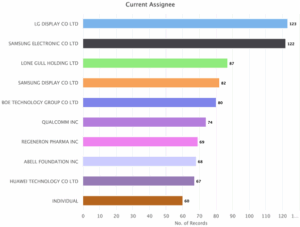
Figure 2: Sea Energy Distribution of patents based on the assignees
As indicated in Figure 2, LG Display CO LTD is the key player with 123 patents followed by Samsung Electronic CO LTD with 122. The next key players are Lone Gull Holding LTD with 87 Patents, Samsung Display CO LTD with 82 patents, Boe Technology Group CO LTD with 80 Patents, Qualcomm INC with 74 patents, Regeneron Pharms INC with 69 Patents, Abell Foundation INC with 68 patents, Huawei Technology CO LTD with 67 patents and individual with 60 patents.
C. JURISDICTION BASED GLOBAL PATENT FILING TRENDS
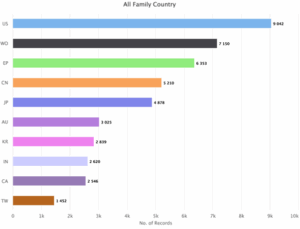
Figure 3: Sea Energy Distribution of patent based on the jurisdiction
Figure 3 shows the distribution of patents based on the jurisdiction. The maximum number of patents i.e., 9042 were filed in the US (United States) and rest of the patents were issued in the other jurisdictions, such as World Intellectual Property Organization (WO) with 7150 patents, European Patent Office (EP) with 6353 patents followed by China (CN) with 5210 patents, Japan (JP) with 4878 patents, Australia (AU) with 3025 patents, Korea (KR) with 2839 patents, India (IN) with 2620 patents, Canada (CA) with 2546 patents and Taiwan (TW) with 1452 patents.
D. TECHNOLOGY CLASSIFICATION BASED ON IPC CLASSIFICATION
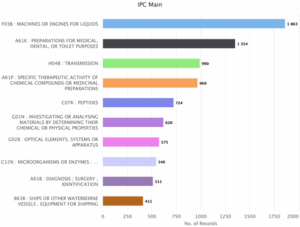
Figure 4: Sea Energy Patent Distribution based on main IPC Code Classification.
Figure 4 indicates the patent distribution based on the main IPC Code. The top spot is occupied by the Machines or Engines for Liquids with 1863 patent families. Other major players include Preparations for medical, dental, Toilet Purposes with 1354 patents; Transmission with 990 patents; Specific Therapeutic activity of chemical compounds or medicinal preparation with 969 patents; Peptides with 724 patents; Investigation or analyzing materials by determining their chemical or physical properties with 620 patents; optical elements, systems or apparatus with 575 patents; Microorganisms or Enzymes with 546 patents; Diagnosis, surgery identification with 511 patents and Ships or other waterborne vessels, equipment for shipping with 411 patents.
E. ACTIVITIES OF COMPETITORS BASED ON AREA OF RESEARCH (IPC)
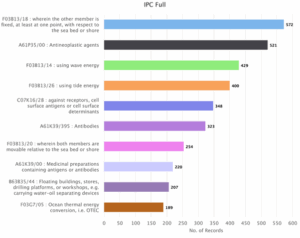
Figure 5: Sea Energy Distribution of Patent based on Competitors and Area of Research (IPC).
Figure 5 depicts the distribution of patents based on competitors and area of research (IPC). The top spot occupied by IPC F03B13/18 i.e., member is fixed at least at one point with respect to the sea or shore holds 572 patents. Other major players includes IPC A61P35/00 i.e., Antineoplastic agents with 521 patents, F03B13/14 i.e., Wave energy with 429 patents, F03B13/26 i.e., tide energy with 400 patents, C07K16/28 i.e., receptors, cell surface antigens or cell surface determinants with 348 patents, A61K39/395 i.e., Antibodies with 323 patents, F03B13/20 i.e., members are movable relative to the sea bed or shore with 254, A61K39/00 i.e., Medicinal preparations containing antigens or antibodies with 220 patents, B63B35/44 i.e., floating buildings, stores, drilling platforms, or workshops, e.g. carrying water-oil separating devices with 207 patents and F03G7/05 i.e., Ocean thermal energy conversion i.e., OTEC with 189 patents.
F. ESTIMATED TOP 10 INDUSTRIES AND TECHNOLOGY AREAS BETWEEN 2014 AND 2024
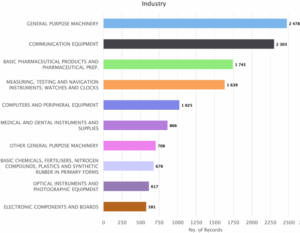
Figure 6: Sea Energy Distribution of Patent based on Industries & Technology.
Figure 6 indicates major industries and technology areas between 2014 and 2024. General purpose machinery is the main industry in the field with 2478 patents. Communication and Equipment follows next with 2303 patents. The next in line is Basic Pharmaceutical Products and Pharmaceutical Prep with 1745 patents, Measuring, testing and navigation instruments, watches and clocks with 1639 patents, Computers and peripheral equipment’s with 1025 patents, Medical and Dental Instruments and Supplies with 866 patents, Other general purpose machinery with 708 patents, Basic chemicals, Fertilizers, Nitrogen Compounds, Plastics and Synthetic Rubber in primary forms with 678 patents, Optical instruments and Photographic equipment with 617 patents and electronic components and boards with 581 patents.
G. ESTIMATED TOP 10 OWNERS BETWEEN 2014 AND 2024
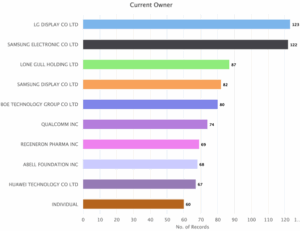
Figure 7: Sea Energy Distribution of Patent based on Current Owner.
As indicated in Figure 7, LG Display CO LTD is the major player with 123 patents followed by Samsung Electronic CO LTD with 122. The next key players are Lone Gull Holding LTD with 87 Patents, Samsung Display CO LTD with 82 patents, Boe Technology Group CO LTD with 80 Patents, Qualcomm INC with 74 patents, Regeneron Pharms INC with 69 Patents, Abell Foundation INC with 68 patents, Huawei Technology CO LTD with 67 patents and individual with 60 patents.
H. ESTIMATED TOP 10 ASSIGNEE COUNTRIES BETWEEN 2014 TO 2024
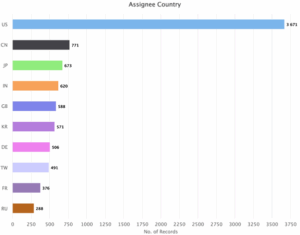
Figure 8: Sea Energy Distribution of Patent based on Assignee Countries.
Figure 8 indicates United states (US) is the key assignee country with 3671 patents followed by China (CN) with 771 patents, Japan (JP) with 673 patents, India (IN) with 620 patents, United Kingdom (GB) with 588 patents, Korea (KR) with 571 patents, Germany (DE) with 506 patents, Taiwan (TW) with 491 patents, France (FR) with 376 patents and Russia (RU) with 288 patents.
I. ESTIMATED TOP 10 PRIORITY COUNTRIES BETWEEN 2014 AND 2024
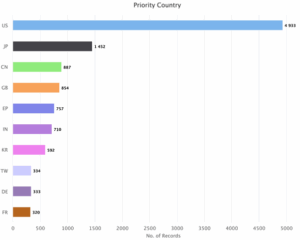
Figure 9: Sea Energy Distribution of Patent based on Priority Countries
As indicated in Figure 9, the United States (US) is the priority country for sea energy with 4933 patent family data set. Japan (JP) is another priority country with 1452 patents. Rest priority countries are China (CN) with 887 patents, United Kingdom (GB) with 854 patents, European Patent Organization (EP) with 757 patents, India (IN) with 710 patents, Korea (KR) with 592 patents, Taiwan (TW) with 334 patents, Germany (DE) with 333 patents and France (FR) with 320 patents.
J. ESTIMATED TOP 10 PUBLICATION COUNTRIES BETWEEN 2014 AND 2024
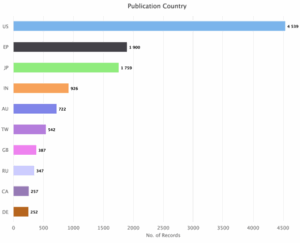
Figure 10: Sea Energy Distribution of Patents based on Publication Countries.
Figure 10 indicates that the maximum patents belong to the investors of United States (US) with 4539 patents and rest patents are issued to the investors of other countries, i.e., European Patent Organization (EP) with 1900 patents, Japan (JP) with 1759 patents, India (IN) with 926 patents, Australia (AU) with 722 patents, Taiwan (TW) with 542 patents, United Kingdom (GB) with 387 patents, Russia (RU) with 347 patents, Canada (CA) with 257 patents and Germany (DE) with 252 patents.
CONCLUSION
The patent landscape analysis for sea energy technologies from 2014 to 2024 reveals a rapidly evolving sector driven by global demand for sustainable energy solutions. The exponential growth in patent filings, particularly peaking in 2023 with over 2,000 applications, underscores intensified research and commercialization efforts in wave, tidal, and ocean thermal energy conversion (OTEC) systems. Leading corporations like LG Display and Samsung Electronics dominate the patent landscape, reflecting cross-industry interest in marine energy applications, while jurisdictions such as the U.S., China, and Japan emerge as key innovation hubs. Notably, fixed marine energy systems and wave energy technologies receive the most attention, whereas OTEC remains relatively underdeveloped, signaling potential growth opportunities. The unexpected overlap with pharmaceutical and electronics patents suggests interdisciplinary advancements, possibly in biofouling-resistant materials or smart energy systems. As governments and industries prioritize decarbonization, this report emphasizes the critical role of strategic IP protection, international collaboration, and targeted R&D investments to accelerate the commercialization of sea energy technologies and unlock their full potential in the global renewable energy transition.
Author: Dashami B.M., PES University, Bangalore
Please contact us at info@origiin.com to know more about our services (Patent, Trademark, Copyright, Contract, IP Licensing, M&A of companies)
Subscribe to YouTube Channel HERE
Join LinkedIn Group: Innovation & IPR
WhatsApp: +91 74838 06607
Jul 4, 2025 | Patent

Wind power is one of the fastest growing sources of power in the world, because of the imperative to clean the air we breathe and the environment we live in. From bigger, more efficient turbines, to smart control systems and offshore solutions, the sector has proved fertile ground for innovation as technology has advanced. In such an environment, a patent landscape report serves as a tool to map out who is developing what and in what specific area and specific manner, which is useful for R&D, business strategy, and investment judgment.
This patent landscape report is designed to identify the trends and innovations in wind energy technology over the years. It will assist those such as researchers, investors, R&D personnel, trend players, policy-makers and companies to find the best and top players, trending segments, emerging growth areas and future demand across the world. It merges technical and strategic explorations involving patent filings, ownership analysis, jurisdictional reach, and technology categorization to offer a targeted perspective on the developments that will shape the future of wind power.
OBJECTIVE OF THE REPORT
This report provides us with an in-depth analysis on global patent activities related to wind energy for a period between the year 2020 and 2025. The aim is to uncover key innovation trends and spot technology and market gaps. The Patent data is supported by technical sources and industry reports for a detailed analysis.
Wind energy is at the forefront of the global transition to clean and reliable power. There is quickly growing innovation in turbines, materials, control systems, and storage. Filing patents has gone up tremendously, indicating substantial R&D and commercial interest globally. However, some critical breakthroughs are necessary to unlock its full potential.
It is also a transitioning energy system, creating economic opportunities. It quickly creates jobs, powers new industries, and cuts emissions. This report serves stakeholders in observing where innovation is happening and where it is not. The goal is simple: map the progress, reveal the gaps, and guide the next move.
METHODOLOGY
This report is based on a comprehensive analysis of patent data related to wind energy technologies from 2020 to 2025. Patent records were sourced mainly from Google Patents and the PatSeer database, forming a set of applications and granted patents. The study includes global filing environment with a concentration on jurisdictions actively engaged in wind-energy innovation.
The patent search strategy was developed using a combination of focused keywords such as “wind energy,” “Wind turbine,” “blade pitch control,” “wind energy storage,” “floating wind platform,” and “digital twin wind turbine.” To narrow down the results to the most relevant innovations, carefully constructed Boolean expressions combined with a set of IPC classifications F03D (wind motors), H02J (power distribution systems), G06F (data processing), and B63B (floating structures) were applied.
Patent families were grouped and duplicates were eliminated; this was done to ensure accuracy in trend mapping. Data were then filtered by applicant type, legal status, jurisdiction, and publication year. Key indicators such as top assignees, priority countries, and technology categories were extracted and analyzed. Visuals were created using various graphical representations to maintain consistency and readability across all parameters. Where necessary, our research has been cross-validated through industry reports, whitepapers, and technical literature for enhanced reliability and context.
This methodology ensures a structured, data-driven approach to understand the evolution of innovation in wind energy during the past decade, thus equipping both technical and strategic stakeholders with valuable insights.
SCOPE
This report provides a comprehensive analysis of wind energy patent activities between 2020 to 2025, with the following defined parameters:
- Geographical coverage : Global, with focused insights on key jurisdictions such as China, USA, Germany, Denmark, India, Japan, and others contributing significantly to wind energy innovation.
- Time period coverage: Patent filings from 2020 to 2025, capturing technological progress and shifts in R&D priorities.
- Patent family coverage : Analysis is based on priority patent families, covering:
- Active and granted patents,
- Pending applications,
- Expired patents for historical insight.
- Technology coverage:
- Onshore and offshore wind turbines, Floating wind platforms,
- Turbine components (blades, towers, gearboxes, generators),
- Control systems (pitch, yaw, SCADA, AI-enabled),
- Grid integration and hybrid systems,
- Energy storage solutions (batteries, hydrogen)
- Sub-technology coverage:
- Blade material innovations (e.g., recyclable composites)
- Floating foundations and mooring systems
- Smart control and monitoring using IoT and digital twins
- Wind-solar hybrid models and microgrids
KEY TECHOLOGICAL AREAS
Wind energy technology today is not just about building turbines, over time it has been evolved from being just turbines to a smarter, more technological connected system. This is clearly visible by the advancement in the digital twin technology which creates a virtual replica of a turbine to simulate real time operational conditions, monitor wear and tear, and optimizes the performance. For example, Siemens Gamesa is working with NVIDIA to develop AI-powered digital twins for its wind turbines.
Alongside there is AI-driven predictive maintenance tools which are used for machine learning to analyse sensor data. This helps in detecting faults, reducing downtime, operational expenses, increases safety and is cost effective. GE’s Predix platform uses machine learning to forecast failures and optimize maintenance schedules. It analyzes terabytes of sensor data across its wind fleets.
Floating wind platforms one of the major developments. It helps in enabling turbine development in deep waters beyond the reach of fixed fountains. Equinor’s Hywind Scotland, built the world’s first commercial floating wind farm and achieved a capacity factor of 54-57% in the first 5 years.
Finally, grid integration technologies include smart inverters and real-time load balancing. All together it shows one thing, that the wind energy technology is becoming more flexible and reliable day by day.
PARAMETER USED FOR THIS ANALYSIS
In this report, we shall study the following parameters:
- Patenting Trends for the year 2022 to 2025.
- Jurisdiction based Global Patent Filing Trends.
- Technology classification based on IPC classification.
- Activities of Competitors based on area of Research (IPC).
- Estimated top 10 industries and technology areas between 2020 and 2025.
- Estimated top 10 owners between 2020 and 2025.
- Estimated top 10 Assignee Countries between 2020 and 2025.
- Estimated top 10 priority countries between 2020 and 2025.
- Estimated top 10 publication countries between 2020 and 2025.
- Distribution of Patents based on their current legal status.
A. Patenting Trends for the year 2020 to 2025
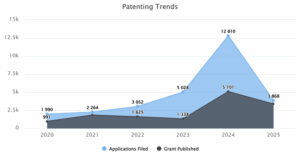
Figure 1: Patenting Trends for the year 2020 to 2025
Figure 1 shows the distribution of patents in wind energy technology from 2020 till 2025. This includes applications filed and grants published. The maximum number of patent applications had been filed in 2024, with figures touching 12,810 filings, followed by 2025, with 3,868 filings. In the case of grants, it was 2024 again when their highest peak was published: 5,101 grants were published, while the lowest number of grants, 991, was in 2020. Up till 2024, patent filings had sharply risen for wind energy, thus depicting a very strong surge of innovation during this period.
B. Jurisdiction based Global Patent Filing Trends
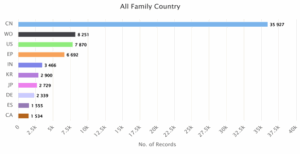
Figure 2: Jurisdiction based Global Patent Filing Trends
Figure 2 shows the global patent filing trends for wind energy technologies based on family country distribution. China leads by a significant margin with 35,927 records, followed by the World Intellectual Property Organization (WO) with 8,251 and the United States with 7,870. The European Patent Office (EP) follows closely with 6,692 records, while India ranks fifth with 3,466. The data reflects China’s dominant role in wind energy innovation, with strong activity also observed in major global and regional patent offices.
C. Technology classification based on IPC classification
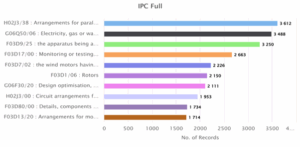
Figure 3: Technology classification based on IPC classification
Figure 3 represents the top IPC classifications by full code, showing specific areas of innovation in wind energy technologies. The most active category is H02J3/38, covering parallel power arrangements, with 3,612 records. This is followed by G06Q50/06 (utility service management) and F03D9/25 (apparatus for wind motors), with 3,488 and 3,250 records respectively. Other key areas include monitoring systems, rotor design, design optimization, and component arrangements, indicating a wide range of innovation focus across both hardware and system-level technologies.
D. Activities of Competitors based on area of research (IPC)
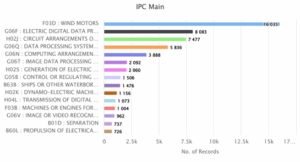
Figure 4: Activities of Competitors based on area of research (IPC)
Figure 4 represents the top IPC main classes associated with wind energy patents, showcasing broader technology categories. In which the dominant class being F03D, covering wind motors, with a significant lead of 16,035 records. Other major areas include electric data processing (G06F) with 8,083 records and circuit arrangements (H02J) with 7,477. The presence of data processing, control systems, and digital transmission categories reflects the growing integration of software, electronics, and automation in modern wind energy technologies.
E. Estimated top 10 industries and technology areas between 2020-2025
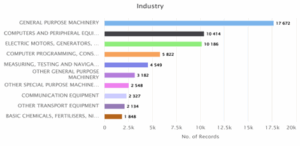
Figure 5.1: Top 10 industries
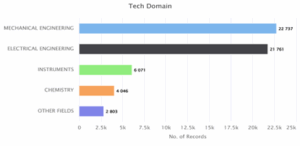
Figure 5.2: Top 5 technological areas
Figure 5.1 gives the number of records in various industries. The General-Purpose Machinery sector tops the list with 17,672 records, followed by Computers and Peripheral Equipment (10,414), and Electric Motors, Generators, and Transformers (10,186). Computer Programming, Consultancy, and Related Activities also contribute significantly with about 5,822 records. The Basic Chemicals and Fertilisers sector has the least, at 1,848. Overall, the data reflects a strong presence of machinery and technology-oriented industries in the record count.
Figure 5.2 illustrates the distribution of records across different technology domains. Mechanical Engineering leads with 22,737 records, followed closely by Electrical Engineering with 21,761, indicating their central role in innovation. Instruments (6,071) and Chemistry (4,046) also make notable contributions. Other Fields having smaller share with 2,803 records. The data highlights a strong concentration in core engineering areas, many of which are critical to advancing wind energy and other emerging technologies.
F. Estimated top 10 owners between 2020 and 2025
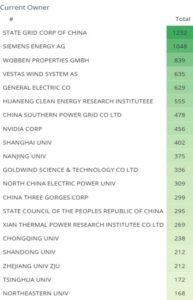
Figure 6: Estimated top 10 owners between 2020 and 2025
Figure 6 shows the top 20 current owners ranked by the total number of patents held by them. State Grid Corporation of China leads with 1,232 patents, followed by Siemens Energy AG with 1,048, and Wobben Properties GmbH with 839. Vestas Wind Systems and General Electric Co. round out the top five with 635 and 629 patents, respectively. The list includes both provate and public sectors such as corporate and academic institutions, reflecting wide-ranging innovation across the wind energy space.
G. Estimated top 10 Assignee Countries between 2020 and 2025
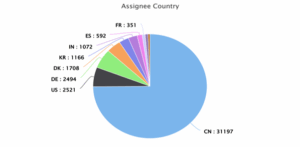
Figure 7: Estimated top 10 Assignee Countries between 2020 and 2025
Figure 7 presents the distribution of wind energy patent assignees by a country. Here again China dominates with about 31,197 assignees, showing its central role in global wind technology development. The United States (2,521), Germany (2,494), and Denmark (1,708) follow from far behind. Other notable contributors include South Korea, India, Spain, and France, underlining a global interest in wind energy innovation.
H. Estimated top 10 priority countries between 2020 and 2025
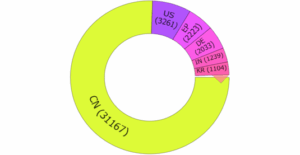
Figure 8: Estimated top 10 priority countries between 2020 and 2025
Figure 8 lists the top priority countries in wind energy patent applications/filings. China holds a commanding lead with 31,167 filings, showing strong domestic innovation and early-stage protection. The United States follows with 3,261, while the European Patent Office (EPO) at (2,223), Germany (2,033), India (1,239), and South Korea (1,104) also feature prominently. These numbers reflect where innovation is first registered and protected, before going onto an international expansion.
I. Estimated top 10 publication countries between 2020 and 2025
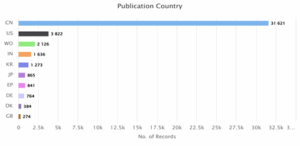
Figure 9: Estimated top 10 publication countries between 2020 and 2025
Figure 9 depicts the distribution of wind energy patents by publication country across the world. China being dominant again with a number of 31,621 published patents, far ahead of the United States at 3,822 and WIPO publications standing at 2,126, followed by India (1,636), South Korea (1,273), and Japan (865). The rest including EP, Germany, Denmark, and the UK have moderate numbers, reflecting broader international engagement but still way behind China’s pace.
J. Distribution of Patents based on their current legal status
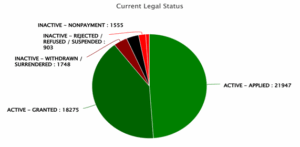
Figure 10:Distribution of Patents based on their current legal status
Figure 10 represents the current legal status of patents in wind energy. The majority fall under active applications (21,947) and active granted patents (18,275), together forming a dominant portion in the given data. In contrast, a smaller share includes inactive patents due to withdrawal or surrender (1,748), nonpayment (1,555), and rejection or suspension (903). This dominant share of active filings indicates ongoing innovation and commercial interest in the wind energy sector.
CONCLUSION
This report captures the rapid evolution of wind energy technologies over the past few years, highlighting the key trends in patent activity, innovation and global participation. The data reflects the growth in filings, especially from countries like China, the United States, Germany, Indian and Korea indicating both commercial interest and policy support for clean wind energy.
The technological advancements is not only on turbines alone, there is growing patent activity in areas such as grid integration, digital twin systems, AI-driven control, floating wind platforms, and hybrid energy solutions. These developments indicates that wind energy is moving beyond mechanical improvements and more toward smarter, more interconnected systems.
While a few major players still dominate the space, it’s encouraging to see more research institutions and new assignees entering the field. This broadening base of innovators opens the door for fresh partnerships, investments, and focused R&D efforts.
In short, the patent landscape report concludes that wind energy is not just scaling in capacity but also in its evolving complexity and capability. For stakeholders across all sectors, this report offers a strategic tool to track progress, identify gaps, and position themselves in a fast-moving global energy market.
Author: Ananya Ainapur , RV University, Bangalore
Please contact us at info@origiin.com to know more about our services (Patent, Trademark, Copyright, Contract, IP Licensing, M&A of companies)
Subscribe to YouTube Channel HERE
Join LinkedIn Group: Innovation & IPR
WhatsApp: +91 74838 06607
Jun 28, 2025 | Patent
This patent landscape report explores global innovation trends in CRISPR (Clustered Regularly Interspaced Short Palindromic Repeats) genome editing technologies, with a specific focus on their application in human therapeutics. CRISPR has revolutionized genetic engineering by enabling precise, efficient, and cost-effective gene modifications. Originally derived from bacterial immune systems, CRISPR—especially the Cas9 variant—has seen widespread adoption in medical, agricultural, and pharmaceutical research.
Focusing on the period from 2014 to 2024, this report examines the exponential growth in patent filings and grants, emphasizing developments in human genome editing, both somatic and germline. It assesses the role of CRISPR in addressing genetic disorders such as cancer, sickle cell disease, and congenital blindness. The analysis includes key metrics such as major assignees, patent jurisdictions, technological domains, and industry sectors.
Beyond quantitative trends, the report delves into qualitative factors shaping the innovation landscape: evolving CRISPR tools like Cas12, Cas13, base editing, and prime editing; major legal battles over intellectual property rights; and the emergence of personalized gene therapies. These insights not only reveal competitive dynamics but also underscore the ethical, regulatory, and societal implications of CRISPR-based innovation. Through this comprehensive analysis, the report aims to map the trajectory of CRISPR technologies and assess their transformative potential in biotechnology and public health.
Objectives
- Examine the global patent filing trends related to CRISPR genome editing in humans from 2014 to 2024.
- Identify key assignees, assignee countries, and priority areas in the CRISPR patent landscape.
- Analyse the main International Patent Classification (IPC) codes associated with CRISPR technologies.
- Assess the distribution of patents across major industries and technological domains.
- To understand the advancements in CRISPR tools (Cas9, Cas12, Cas13, base editing, prime editing) and their impact on innovation.
- Evaluate real-world applications and ethical considerations through notable case studies like Baby KJ.
- Explore the implications of patent disputes and licensing on future CRISPR-based therapies and global research efforts.
What is Human Genome Editing?
Human Genome Editing refers to changing DNA in a specific way. It can add, remove, or fix DNA in the genome. It can be applied in 3 ways – somatic cells (not passed to children),germline cells(not for reproduction), germline cells (for reproduction-heritable).
 The application of somatic human genome editing has been undertaken, including in vivo editing ,to address HIV and sickle-cell disease , for example. Even though somatic human genome editing is accepted in many countries, and they are many benefits, challenges still preside. One major challenge is creating innovations that consider the diversity of the human population and experience.[1] Other challenges that preside are medical travel , unethical clinics and the reporting of illegal, unregistered, unethical, or unsafe research.
The application of somatic human genome editing has been undertaken, including in vivo editing ,to address HIV and sickle-cell disease , for example. Even though somatic human genome editing is accepted in many countries, and they are many benefits, challenges still preside. One major challenge is creating innovations that consider the diversity of the human population and experience.[1] Other challenges that preside are medical travel , unethical clinics and the reporting of illegal, unregistered, unethical, or unsafe research.
Whereas heritable human genome editing is referred when nuclear DNA is edited in a way that is heritable across generations. However, this type of editing has be severely debated over in regard to the possible consequences for offspring and for society in general. It may also cause safety and ethical issues.
Therefore, somatic human genome editing is mostly preferred when compared to heritable human genome editing.[2]
What is CRISPRR Genome Editing ?
The National Human Genome Research Institute states CRISPR (clustered regularly interspaced short palindromic repeats) to be “ a technology that aids researchers in the selective modification of the DNA of living organisms. CRISPR was adapted for laboratory use from naturally occurring genome editing systems found in bacteria.”[3]
CRISPR has been used by scientist to add, remove or change parts of DNA to fix problems and understand diseases presiding in the DNA.
Currently clinical test is being undertaken revolving CRISPR especially regarding cystic fibrosis, sickle cell disease, cancer, and HIV. There are hopes that CRISPR will help aid and provide treatment for these major illnesses.
A natural defence system in bacteria has also led to another major invention CRISPR-Cas9. It is currently used as a special guide for RNA so that it can find the exact spot in the DNA to cut. The Cas9 enzyme is then used to cut the DNA and make the required modifications.[4]
There have also been some ethical concerns such as if the modified DNA would be continued by future changes and the effects presiding in embryos, sperm, and egg cells. Due to these challenges some countries have been reluctant to allow human genome editing in their country.[5]
What is CRISPR -Cas9 ,Cas12,Cas13?
CRISPR-Cas9 can be considered as a pair of scissors it cuts DNA and makes the required modifications. It identifies a particular disease and either adds, changes or removes something in the DNA to solve the problem. The changes made by this type of technology remain permanent and preside in the body forever. However, it should be noted that the technology can sometimes also cut the wrong part of the DNA which can cause major problems.
CRISPR -Cas12 can also be used to modify DNA, however it makes staggered cuts instead of straight cuts like CRISPR- Cas9. It can also cut single-stranded DNA nearby. This also aids in finding major viruses and diseases. It is also considered to be much easier to use than the other types of CRISPR technologies. It is useful in research and testing as it can modify many genes at once.
CRISPR-Cas13 is different from the other two types of technologies as it makes changes to RNA instead of DNA. It prevents the creation of disease making proteins. Therefore, the changes are only considered temporary as changes are to RNA instead of DNA.
In summary, Cas9 and Cas12 are used for permanent changes in DNA, while Cas13 is used for temporary changes in RNA. Scientists choose between them based on whether they want to change DNA forever or just temporarily stop harmful effects.
Parameters used for Patent Landscape analysis.
- Patent filing trend between years 2014 to 2024
- Distribution of Patents based on the Assignees.
- Jurisdiction based Global Patent Filing Trends
- Technology classification based on IPC classification.
- Activities of Competitors based on area of Research (IPC)
- Estimated top ten industries and technology areas between 2014 and 2024.
- Estimated top 10 owners between 2014 and 2024.
- Estimated top 10 Assignee Countries between 2014 and 2024
- Estimated top 10 Priority Countries between 2014 and 2024
- Estimated top 10 Publication Countries between 2014 and 2024
Patent filing trend between years 2014 to 2024
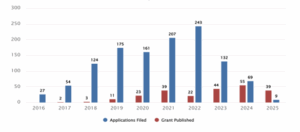 Figure 1 : Patent filing trend between the years 2014 to 2024.
Figure 1 : Patent filing trend between the years 2014 to 2024.
Figure 1 illustrates the trend in patent activity related to CRISPR human genome editing from the year 2014 to 2024, covering both applications filed and grants published. A steady increase in patent filings is observed from 2016, peaking in 2022 with 243 applications, followed by a decline in 2023 (132) and 2024 (69). The number of patents granted remained low in the early years, with only 2 to 3 grants annually between 2016 and 2018. However, a notable rise occurred from 2020 onwards, with the highest number of grants (44) published in 2023, followed by 39 in 2021 and 22 in 2022. While 2022 saw the most applications filed, the highest number of grants was recorded in 2023, indicating a time lag between filing and grant. Overall, the data suggests a significant surge in innovation and interest in CRISPR genome editing technologies, particularly between 2018 and 2022, followed by a slight decline post-2022.
Distribution of Patents based on the Assignees.
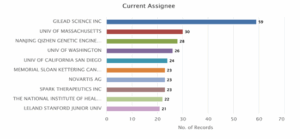
Figure 2 presents the distribution of current assignees for patents related to CRISPR human genome editing. Gilead Sciences Inc. leads significantly with 59 patent records, indicating a dominant role in the field. This is followed by the University of Massachusetts with 30 patents and Nanjing Qizhen Genetic Engineering Co. Ltd. with 28 records. Other notable contributors include the University of Washington (26) and University of California San Diego (24). Several institutions such as Memorial Sloan Kettering Cancer Centre, Novartis AG, and Spark Therapeutics Inc. each hold 23 patents, highlighting strong industry and academic involvement. The National Institutes of Health (22) and Stanford University (21) also feature among the top assignees. Overall, the chart highlights a balanced mix of academic, public, and private sector innovation in advancing CRISPR genome editing technologies.
Jurisdiction based Global Patent Filing Trends
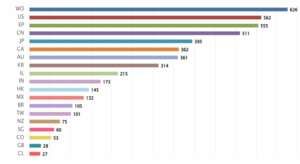
Figure 3: Distribution of Patent based on the jurisdiction.
Figure 3 shows the distribution of patents based on the jurisdiction in which they were filed. The highest number of patents were filed under the World Intellectual Property Organization (WO) with 626 applications, followed by the United States (US) with 562 patents and the European Patent Office (EP) with 555 patents. China (CN) and Japan (JP) also contributed significantly, with 511 and 395 patents, respectively. Other notable jurisdictions include Canada (CA) with 362, Australia (AU) with 361, and South Korea (KR) with 314 patents.
Technology classification based on IPC classification.
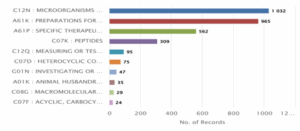
Figure 4: CRISPR Genome Editing in Humans Patent Distribution based on main IPC Code Classification
Figure 4 displays the distribution of patents across different International Patent Classification (IPC) codes relevant to CRISPR human genome editing. The highest number of patents fall under C12N: Microorganisms or Enzymes, with a total of 1,032 records, indicating a strong focus on genetic manipulation and biotechnology. This is closely followed by A61K: Preparations for Medical, Dental or Toilet Purposes with 965 records, and A61P: Specific Therapeutic Activity of Chemical Compounds or Medicinal Preparations with 562 records, both highlighting the medical and therapeutic applications of CRISPR. Other significant IPC codes include C07K: Peptides (309) and C12Q: Measuring or Testing Processes Involving Enzymes or Microorganisms (95). Categories like C07D: Heterocyclic Compounds (75) and G01N: Investigating or Analysing Materials (47) also appear prominently. Lesser but notable classifications include A01K: Animal Husbandry (35),
C08G: Macromolecular Compounds (29), and C07F: Acyclic, Carbocyclic Compounds (24). This data indicates a diverse yet concentrated research focus, primarily in medical, biochemical, and microbiological domains.
Activities of Competitors based on area of Research (IPC)
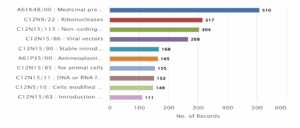
Figure 5: CRISPR Genome Editing in Humans – Patent based on Competitors and Area of Research (IPC)
Figure 5 illustrates the IPC Full-level classification of patents related to CRISPR human genome editing, highlighting the specific technologies being targeted. The leading IPC category is A61K48/00: Medicinal preparations containing genetic material with 510 records, reflecting the dominant focus on gene-based therapies. This is followed by C12N9/22: Ribonucleases (317) and C12N15/113: Non-coding nucleic acids (304), indicating extensive research into RNA-related technologies and genetic regulation mechanisms. Other significant areas include C12N15/86: Viral vectors (268) used for gene delivery, and C12N15/90: Stable introduction of genetic material into eukaryotic cells (168). Additionally, A61P35/00: Antineoplastic agents (165) show the therapeutic targeting of cancer using CRISPR. Categories such as C12N15/85 (for animal cells – 155), C12N15/11 (DNA or RNA fragments – 152), and C12N5/10 (modified animal cells – 149) further demonstrate the cellular and molecular focus of these innovations. Finally, C12N15/63: Introduction of foreign genetic material using vectors with 111 patents emphasizes the technical approach in CRISPR delivery mechanisms. Overall, the graph confirms that gene therapy and genome editing applications form the core of CRISPR-related research.
Estimated top 10 industries and technology areas between 2014 and 2024.
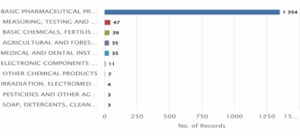
Figure 6: CRISPR Genome Editing in Humans – Distribution of Patent based on Industries & Technology.
Figure 6 illustrates the distribution of patents across major industries and technology areas from 2014 to 2024 in the domain of CRISPR human genome editing. The dominant industry by a large margin is Basic Pharmaceutical Products and Preparations, with 1,354 patent records, indicating the primary application of CRISPR in drug development and therapeutics. This is followed by Measuring, Testing and Navigating Equipment with 47 records, and Basic Chemicals, Fertilisers, and Nitrogen Compounds with 39 records. Other notable sectors include Agricultural and Forestry Machinery (35), Medical and Dental Instruments (35), and Electronic Components and Boards (11). Smaller contributions come from industries such as Other Chemical Products (7), Irradiation and Electromedical Equipment (4), and Pesticides and Other Agrochemical Products (3). The Soap, Detergents, and Cleaning Products industry recorded the least number of patents, with just 3 records. Overall, the graph emphasizes the biopharmaceutical and healthcare sector’s dominance in CRISPR-related innovations, with limited but emerging applications across agriculture, electronics, and chemical industries.
Estimated top 10 owners between 2014 and 2024.
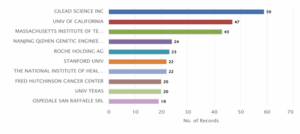
Figure 7: CRISPR Genome Editing in Humans – Distribution of Patent based on Current Owner
Figure 7 highlights the key assignees based on the number of patent records. Gilead Science Inc. emerges as the leading assignee with 59 patents, followed by the University of California with 47 and the Massachusetts Institute of Technology with 43.
Other significant contributors include Nanjing Qizhen Genetic Engineering Co. Ltd. with 24 patents, Roche Holding AG with 23, and both Stanford University and The National Institute of Health with 22 each.
Additionally, Fred Hutchinson Cancer Centre and University of Texas have each filed 20 patents, while Ospedale San Raffaele SRL rounds out the top assignees with 19 records.
Estimated top 10 Assignee Countries between 2014 and 2024
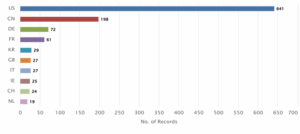
Figure 8: Distribution of Patent based on Assignee Countries
Figure 8 displays the distribution of patents based on the assignee countries. The United States (US) holds the highest number of records with 641 patents, making it the leading jurisdiction. It is followed by China (CN) with 198 patents and Germany (DE) with 72.
Other significant contributors include France (FR) with 61 patents, South Korea (KR) with 29, and both the United Kingdom (GB) and Italy (IT) with 27 each. Ireland (IE) and Switzerland (CH) recorded 25 and 24 patents respectively, while Netherlands (NL) accounted for 19.
Estimated top 10 Priority Countries between 2014 and 2024
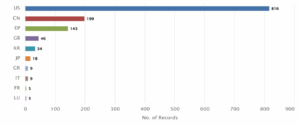
Figure 9: Distribution of Patent based on Priority Countries
Figure 9 shows the distribution of patents based on the priority country. The United States (US) holds the largest number of priority filings, with 816 patent records, followed by China (CN) with 199 and the European Patent Office (EP) with 143.
Other key priority jurisdictions include the United Kingdom (GB) with 46, South Korea (KR) with 34, and Japan (JP) with 18. Meanwhile, Greece (GR) and Italy (IT) contributed 9 patents each, while France (FR) and Luxembourg (LU) had 5 each, representing the lower end of the distribution.
Estimated top 10 Publication Countries between 2014 and 2024
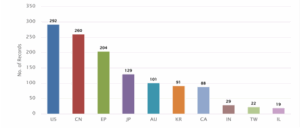
Figure 10 : Distribution of Patent based on Published Countries
Figure 10 represents the distribution of patents based on the inventor’s country. The United States (US) accounts for the highest number of records, with 292 patents, followed closely by China (CN) with 260 and the European Patent Office (EP) with 203.
Other significant contributors include Japan (JP) with 128 patents, Australia (AU) with 100, South Korea (KR) with 91, and Canada (CA) with 88 records.
Further, India (IN) accounts for 29 patents, followed by Taiwan (TW) with 22 and Israel (IL) with 19, representing the lower range of inventor country contributions.
CRISPR and Human Health
CRISPR aims to eliminate diseases caused by human genes it does this through many means :
- Blood Disorders– Scientist identify the faulty cells and take them to the lab for modification. After the needed modifications are made, they are returned into the patient’s body. Results have proved to be effective.
- Blindness – In 2020 ophthalmologists used CRISPR for the first time to cure blindness in their patients. They inject CRISPR directly in the eye to find the rare blindness causing gene and remove it. Ophthalmologists say if this proves to be a safe means they will continue using it.
- Cancer – Doctors remove the cancer-causing cells , CRISPR is then used to make the cells have a better chance at fighting the cancer. The cells are then kept back in the body. It was found that the cancer didn’t grow as much, and the treatment was safe.
- Fast Diagnosis – CRISPR is used to find viruses and diseases in a very quick manner. It helped create the first FDA-approved COVID-19 test using CRISPR.
CRISPR is still being tested, but it offers hope for treating many diseases—from rare genetic conditions to common illnesses like cancer and viral infections.[7]
CRISPR Modifications and Advancements
CRISPR technology has evolved over time with the system being more accurate, faster, and efficient in detecting human genome diseases. The major upgrades are :
- Modified Cas9 Proteins – Cas9 is used to add , remove or change DNA and detect diseases. Recently, scientist have made new inventions such as nCas9 (nickase Cas9) which only cuts one strand of DNA. dCas9 (dead Cas9) instead of cutting DNA it binds it. These two strategies have been proven to be a safer means of human genome editing. This can be attached to editing enzymes to change DNA without breaking it.
- Base Editing – This uses nCas9 or dCas9 + adeaminase enzyme and can change one letter in DNA for example “A to G” etc. This method has proved to be effective as there is no double- strand breaks. However, one limitation is that not all DNA can be edited.
- Prime Editing – This has proven to be a newer and more effective means to human genome editing. It uses nCas9+ reverse transcriptase+ special guide RNA (pegRNA).It is known to correct more types of mutations and correct 89% of human genetic mutations.
- RNA Editing – It uses Cas13b+ adenosine deaminase. It edits RNA and therefore is much safer because the edits are not permanent. It is useful for diseases where temporary fixes are enough.
- Easier Delivery Systems –To treat people as these tools enter the cells and are often used to detect major diseases like AAV The initial problem was that Cas9 is too big for AAV. The solution was a smaller enzyme called AsCas12f which is 1/3 of Cas9. This proved fit inside AAV and was proved to successfully when assessed on mice.
- Why This Matters for Human Health
- Fewer mistakes when editing genes.
- More types of genetic diseases can be treated.[8]
- Better delivery into the human body.
- Safe and specific therapies can be developed for individual patients.
CRISPR Patent Dispute
The discovery of CRISPR- Cas9 has led to a major patent battle between 2 parties. One of the major reasons for the dispute was financial stakes and who would have authority over the technology especially regarding human cells.
The two parties were Jenifer Doudna from the University of California, Berkely and Emmanuelle Charpentier from the University of Vienna & Umeå University who filed their patent in May of 2012.
The other party was Feng Zhang from the Broad Institute of MIT and Harvard. He filed his patent in December of 2012,just as his paper showing CRISPR use in human cells was accepted.
By April 2014 Zheng’s team was granted the first patent while Doudna’s was still under review. This led to Doudna’s team challenging the decision.
Eventually by 2019 both parties were granted patent access, biotech companies would either get licenses from Doudna or Zhang.
However, in February of 2022 the U.S Patent Office sided with Zhang. This cause major financial problems for Doudna’s team.
It should be note that in Europe and other countries Doudna and Charpentier won and hold major patents.
- The patent controls who can profit from using CRISPR for human gene editing.
- It affects:
-
- Biotech companies creating CRISPR-based therapies.
- Patients waiting for treatments.
- Global research and licensing costs.[9]
Current News – The case of Baby KJ
The Children’s Hospital of Philadelphia and Penn Medicine hit a major milestone in February 2025 by using CRISPR to treat a baby named KJ. KJ was born with a rare genetic condition called CPS1 deficiency. The condition stops the body from breaking down harmful ammonia and if left untreated it cause major brain and liver damage.[10]
Due to KJ’s young age a liver transplant was not feasible, doctors created a personalised CRISPR therapy accustomed to him .
They used base editing for the process to correct the faulty gene present in his liver. his therapy was delivered through lipid nanoparticles and given in three doses by April 2025.
KJ had responded well:
- He could eat more protein without harmful effects.
- He needed fewer medications.
- He recovered from childhood infections without serious complications.
- No serious side effects had occurred.
The treatment marked the first CRISPR had been personalised, it showed CRISPR could go beyond common diseases and could tackle unique or ultra-rare mutations.
Doctors hope that this success can pave the way for more personalised CRISPR treatments for many diseases that currently have no cure.
However, long-term follow-up is still needed to see how well it works overtime.
Analysis of Report
The patent landscape analysis clearly shows a significant surge in CRISPR-related innovation between 2014 and 2024, particularly from 2018 to 2022. The peak in applications in 2022 and the rise in grants by 2023 indicates both growing research interest and maturing technologies ready for real-world use.
Key players such as Gilead Sciences Inc., the University of California, and the Massachusetts Institute of Technology dominate the patent filings, suggesting strong competition between academic institutions and private companies. The data also shows that countries like the United States, China, Japan, and Germany are leading both in terms of assignee location and inventor nationality, underlining their role as global innovation hubs in biotechnology.
From an industrial perspective, Basic Pharmaceutical Products and Preparations hold the largest number of patents, confirming that CRISPR is primarily being applied in drug development and treatment of genetic diseases. The IPC classification further reflects this, with a major focus on gene therapy, viral vectors, and non-coding nucleic acids.
The analysis also highlights technological diversification with the evolution of CRISPR tools like Cas9, Cas12, Cas13, base editing, and prime editing, all of which aim to increase precision and safety. These advancements are essential in addressing ethical and clinical concerns, particularly in sensitive areas like embryo editing.
Moreover, real-world applications such as the case of Baby KJ demonstrate how CRISPR is transitioning from experimental use to personalised medicine, offering hope for rare and otherwise untreatable conditions.
However, the report also points to ethical debates, regulatory inconsistencies, and intellectual property disputes that may shape or limit access to CRISPR-based treatments. The patent conflict between Doudna and Zhang reflects the high commercial stakes and the complexity of navigating ownership in a rapidly evolving field.
Conclusion
CRISPR genome editing has shown rapid growth between 2014 and 2024, with a strong focus on pharmaceutical and medical applications. The rise in patent filings and technological advancements like Cas9, Cas12, and prime editing reflect its potential in treating genetic disorders.
While countries like the US and China lead in innovation, ethical concerns, patent disputes, and global regulations remain key challenges. Real-world cases, such as Baby KJ, highlight the shift towards personalised CRISPR therapies, offering hope for rare and serious conditions.
Overall, CRISPR stands as a powerful tool in modern medicine, but its future depends on responsible innovation and global cooperation.
Disclaimer
This landscape report is based on resources available in public domain such as published patent applications and granted patents freely available/accessible/downloadable generally paid and Government based patent databases unless specified otherwise. Moreover, the accuracy of the records kept and maintained by the respective patent offices and registries. As such, we are not responsible for any error, omission, and mistake or misfiled particulars contained or omitted from the said records. It must also be noted that the said records may not be updated on a regular basis.
Furthermore, the report is based upon individual expert’s view/judgment. Such analysis may vary from expert to expert. Experts’ views may not concur with/should not be treated as the views of the Author.
Author: Ishika Sarah Koshy , REVA University, Bangalore
Please contact us at info@origiin.com to know more about our services (Patent, Trademark, Copyright, Contract, IP Licensing, M&A of companies)
Subscribe to YouTube Channel HERE
Join LinkedIn Group: Innovation & IPR
WhatsApp: +91 74838 06607
Jun 20, 2025 | General, Patent
The Unitary Patent System was created to make patent protection in the EU easier and cheaper. Before this, inventors had to apply separately in each country. Now, one application gives protection in many EU countries. The system also introduced the Unified Patent Court (UPC), which handles disputes in a central way. This change helps inventors save money and avoid legal confusion. However, the system still has problems, such as high costs, court complexity, and confusing overlap with national and European patents. This paper looks at both the benefits and challenges of the new system.
Unitary Patent System
The Unitary Patent System(UPS) established on June 1st, 2023, has created a significant shift in the process of patenting in the EU. The system has introduced a single valid patent across multiple EU Member states therefore reducing the administrative and financial risk associated with obtaining patent protection in Europe.
 Previously, applications would be validated and maintained through separate patents, meaning that each member state had to grant patent following the provisions established under the European Patent Office (EPO) . However, this process was long and strenuous including translation requirements, validation formalities and the appointment of local patent attorneys. This also led to the of protection for inventions to only a few jurisdictions.
Previously, applications would be validated and maintained through separate patents, meaning that each member state had to grant patent following the provisions established under the European Patent Office (EPO) . However, this process was long and strenuous including translation requirements, validation formalities and the appointment of local patent attorneys. This also led to the of protection for inventions to only a few jurisdictions.
The system has held the inventors to obtain unitary protection by a single legal process . This has broadened the geographic scope and cost-effectiveness of patent coverage in Europe.
This reform is expected to strengthen the EU’s position in global innovation, foster cross-border collaborations, and promote research, development, and industrial investment.
Benefits of UPS
- More innovation as patent protection has a wider geographic scope
- More competition amongst businesses which therefore leads to a reduction in prices
- More job opportunities and a stronger economy
- As inventor’s receive patent protection there is a surety for customers that the products have a good quality assurance [1]
What is a Unitary Patent
A unitary patent is a single legal patent that allows EU Member States that have agreed to the agreement to apply in one place for patent protection instead of going to a long and strenuous process of applying for patent protection in each EU state.
Right now, 18 EU countries are part of this system. These are: Austria, Belgium, Bulgaria, Denmark, Estonia, Finland, France, Germany, Italy, Latvia, Lithuania, Luxembourg, Malta, the Netherlands, Portugal, Romania, Slovenia, and Sweden. More EU countries may join in the future. But non-EU countries (like the UK, Switzerland, or Turkey) cannot be part of the unitary patent.
The unitary patent is based on two EU laws and an international agreement (the UPC Agreement) signed by these EU countries. This is based on the European patent granted by the European Patent Office.
Since 2023 an owner can apply for a patent grant in 2 ways:
- By a unitary patent wherein the patent is granted in all 18 EU Member States (that have agreed to the agreement)
- By using the old system where the owner would have to receive permission from each country – this is called “bundle” of national patents . Countries like UK and Spain which have not agreed to the agreement use this.
The process to apply for and examine a European patent at the EPO stays the same. It’s only after the patent is granted that you make the choice.
This means a patent owner can:
- Use the unitary patent for the 18 EU countries that joined, and
- At the same time, validate the same European patent in other countries separately.[2]
Advantages of Unitary Patents
The main goal of Unitary Patents was to make patent protection easier and cheaper. It especially focused on encouraging more innovation in Europe.
- Avoids the long and strenuous process of applying for patent protection in each EU state for patent grant
- No need to pay renewal fee separately in each country
- No need for multiple translations – Only English or one EU official language
- No extra fee for the unitary request- normal EPO application fees are enough[3]
History of Unitary Patent System
In 1978, the European Patent Convention (EPC) stated the importance of patent protection in Europe. Around the same time discussion about a Community Patent was being discussed wherein a single court could grant patent protection to all EU inventions .However this implementation was not enforced.
In 2004, EU Member states could not agree in terms of language rules for the new system. This also led to the idea of the community patent being dropped.
By 2010,a new proposal for the unitary patent system was enforced. It also stated a special court should be established. By 2012 the member states agreed on the creation of a unitary patent and the language to be used , only Spain and Italy choose not to join at this stage.
By 2013,all Member States except Spain and Poland signed the agreement for a Unified Patent Court, which would handle both unitary patents and traditional European bundle patents. For the system to be enforced 13 Member States had to agree to the proposal.
In 2015,Spain had challenged the regulations, but this was rejected by the European Court of Justice . Italy had agreed to join the system. More countries had started ratifying the agreement including the UK.
In 2017, Hungary decided to drop out of the system as the system was not in accordance with its constitution. Germany had filed a legal case challenging the system as the president was told not to sign an agreement until a resolution was found.
In 2020, UK due to Brexit stated it would not join stating that the country wanted legal independence. In March, the German Constitutional Court said the original vote to approve the UPC was invalid due to the lack of members. A new vote happened in November and December 2020, and this time it passed with the required majority.
Germany had made two final legal complainants , however in 2021 this was rejected by the German Court.
By 2022, Austria became the 13th country to approve the Protocol on Provisional Application . This allowed the system to begin preparations, like setting up the legal framework, IT systems, and hiring judges.
In 2023, Germany had officially ratified the agreement. The UPC started working on 1st June 2023, and the unitary patent system officially began.[4]
Key Differences from Unitary Patent System from the traditional European Patent System
| Feature |
Unitary Patent |
Classic European Patent |
| Geographical Scope |
One patent covers up to 25 EU countries (currently 17 ratified) |
Must be validated individually in each EPC country |
| Application Process |
Single application at EPO, followed by a request for unitary effect |
Application at EPO, then validation needed in each desired country |
| Translation Requirements |
Fewer translations required (cost saving) |
Expensive due to multiple translations and local legal requirements |
| Cost |
Lower initial costs due to centralization and fewer translations |
Higher costs due to country-by-country validation and legal representation |
| Maintenance Fees |
Single renewal fee for all covered states |
Pay separate renewal fees in each country where validated |
| Flexibility in Dropping Markets |
No flexibility—must maintain all or none |
Can drop individual countries to save on fees |
| Legal Enforcement |
UPC has central jurisdiction (enforcement and validity decisions apply across all member states) |
Legal action and invalidation handled separately in each country |
| Risk of Invalidation |
One ruling by UPC can invalidate protection in all covered countries |
A ruling in one country does not affect others |
| Licensing |
Uniform treatment—no regional licensing flexibility |
Can license rights differently across countries |
| Judicial System |
UPC is new and untested (may have unpredictability) |
National courts like in France or Germany are experienced in patent law |
| Best For |
SMEs seeking cost-effective, simple protection across many markets |
Large corporations, especially in pharma and high-stakes industries seeking strategic flexibility and reduced invalidation risks[5] |
How does the Unitary Patent System Work ?
New Strategic Choices for Patent Owners
Since the enforcement of the system in 2023 patent owners now must make two important decisions during the time of accessing patent grants in Europe.
The first decision is choosing between a unitary patent or national validations. Unitary patents mean that the patent owner does not have get separate patent protection from each country. The other way is applying for patent protection separately in each country. It should be noted one cannot use both methods for the same country for example if the patent owner chooses national validation, they cannot also choose unitary patents in the same country. For other European countries that are not in the unitary patent system, you can still validate the patent nationally, no matter what you choose.
It should also be noted that once one decides to opt for a unitary patent they cannot out of the UPC. This in turn means the UPC must be involved in the process of obtaining an unitary patent. One must also use the UPC for national patents in countries that join the UPC in the future.
The next major decision is regarding “opting out” of the UPC . This decision typically arises when one chooses to from national validation instead of a unitary patent . If the patent owner does not opt out the European patent falls under the UPC once granted.
Typically, a patent owner will choose to opt out , especially when important patents are present, to avoid the risk of losing the patent in many countries at once.
The UPC is a new court that covers a wide range of countries, and its decisions can be implemented in each country as well. However, it should be noted that one legal case can cancel the grant of patent in all UPC countries . Also, as the system is new the decisions can tend to be less predictable. That is why some owners choose to opt out their valuable patents from the UPC system.[8]
Filing an Opt-out
As per Article 83(3) of the Agreement on a Unified Patent Court (UPCA) [9]an opt out is when a patent is removed out of the UPC system. This is typically done if someone has a classic European Patent or a Supplementary Protection Certificate (SPC) based on a classical patent. Once opted out, the UPC will not have any authority over legal cases related to that patent.
Conditions for a valid opt-out:
- All countries covered by the patent must be included in the opt-out.
- You can only opt out before any legal case has started in the UPC about that patent.
- Opt-outs must be filed through the UPC’s Case Management System (CMS), following Rule 5 and Rule 5A of the UPC’s Rules of Procedure[10].
- If you’re filing many opt-outs at once, you can use an API for secure bulk submission.
Can you change your mind?
One can withdraw the option to opt out if no case had been started in a national court regarding the patent. One can also correct a mistake , remove an unauthorised opt-out, or withdraw an unauthorised withdrawal through the CMS.
Unified Patent Court
Previously if a patent owner wanted to receive a patent grant in Europe, they would have to receive permission from each country the wanted to receive the grant from, for example a decision by a French court on the validity or infringement of a particular European patent currently has no effect in Germany. This created “parallel litigation ” which can in turn increased costs for the parties concerned as well in resulting in inconsistent decisions from different courts.
The countries signed up to the Unified Patent Court have all agreed to transfer jurisdiction over European patents and SPCs from their national courts to a single, centralised court.
The emergence of the UPC had caused a significant decrease in parallel litigation and inconsistent due to its enforcement of a single court , to hear patent disputes.
What will be litigated in the Unified Patent Court?
- Since its establishment all matters concerning unitary patents have been handled by the UPC . Within contracting states, the UPC has the right to hear matters concerning traditional patents, especially in relation to jurisdiction, validity and infringement . This also includes matters revolving around SPC’s.
- Due to the UPC agreement any matter concerning revocation will now be undertaken by the UPC instead of France. This also involves any infringement of European patents in France.
- It should be noted that during the transition period (, patent matters could still be dealt by the national courts. Also, it should be noted that any patent owner can “opt out” of the UPC during this time .
- National patents however will always be litigated by national courts.
Advantages and Disadvantages of the Unified Patent Court
| Advantages |
Disadvantages |
| Single litigation |
Due to the UPC being a new establishment there could be the result of unpredictability in the initial years and limited precedents |
| A central revocation |
Bifurcation due to the differences in hearings regarding patent validity and infringement |
| A wide scope of experienced judges from Europe |
Forum Shopping
· Some parts of the court may seem to favor patentees more than others.
· People may choose where to file based on where they think they’ll win.
· But as the Court of Appeal makes more decisions, things should become more uniform across divisions.[12]
|
Current Challenges
In 2023 the establishment of a new kind of patent emerged called the unitary patent emerged. This patent would reduce the burden of patent owners regarding receiving patent grants in Europe, they would no longer have to receive permission from each country to get patent instead they would have to appear to the UPC to receive grants. The patent covers 25 countries in the EU. The system was also enforced to decrease parallel litigation and inconsistencies in decisions amongst different courts.
Currently the grant of a European patent also presents to be a challenge regarding translations and the difference of cost between countries , the UPC helped reduced this.
However, there are still some major problems:
- There are too many patent system (National patents, European patents and unitary patents) .This makes it confusing for patent owners.
- Even the UPC has cut the translations cost the annual renewal fees remains high .It could cost around €40,000 to €50,000 for 10 years – which is much more than in the USA or Japan. Even for small firms, the cost might still be around €20,000, which is too high.
- Complicated Court System- The court system for patent disputes will be split between France, Germany, and the UK (before Brexit).This could lead to forum shopping, where companies choose the court that favours them.[13]
Conclusion
The Unitary Patent System is a major improvement for inventors in Europe. It offers simpler, faster, and wider patent protection, which can help encourage innovation. The Unified Patent Court also helps reduce legal delays and conflicting decisions. However, the system still has drawbacks, like expensive renewal fees, unclear legal rules, and too many overlapping systems. These issues make it hard for small companies to fully benefit. For the system to succeed, it should become more affordable, clearer, and better organised. Solving these problems will make the UPS more effective and attractive to users across Europe.
Author: Ishika Sarah Koshy , REVA University, Bangalore
Please contact us at info@origiin.com to know more about our services (Patent, Trademark, Copyright, Contract, IP Licensing, M&A of companies)
Subscribe to YouTube Channel HERE
Join LinkedIn Group: Innovation & IPR
WhatsApp: +91 74838 06607
Mar 7, 2025 | Patent
Compulsory licensing refers to a legal means by a government that permits a third party to make, use, or sell a product or invention without the consent of the patent owner. The patent owner retains their patent rights, but they are entitled for a renumeration for the use. This is an exception to the general rule that only the patent owner has the authority to control the use of their invention.
 The legal framework for compulsory licensing are established through international agreements such as [1]TRIPS Agreement and the [2]Paris Convention, as well as in the domestic laws of the countries.
The legal framework for compulsory licensing are established through international agreements such as [1]TRIPS Agreement and the [2]Paris Convention, as well as in the domestic laws of the countries.
Since compulsory licensing limits the intellectual property rights of an individual, it is considered as a serious measure and may lead to different opinions about the fairness or advantages for the economy. In rare cases, if a company with substantial market power refuses to share its technology, a competition authority may intervene and mandate the issuance of the license to others because keeping it to themselves would be unfair.[3]
Compulsory License of patents In India
Legal Provisions
Section 84 of the Indian Patents Act,1970 [4]–
- Any interested party can appear to the Controller ( Patent Authority) after 3 years from the date a patent is granted, to apply for a compulsory license for the invention if:
- The needs of the public are not being met.
- The product is not available at a reasonable price.
- The invention is not being used in India.[5]
- An individual who already has a patent license can also appear for compulsory stating the reasons as mentioned in subclause. (1)
- The application must include the applicant’s interest and the required facts supporting the claim.
- The controller will grant a license is he agrees with the conditions stated in subclause. (1)
The Controller may also use the provisions in Section 88 (Powers of Controller in granting compulsory licences)[6] while ordering the patentee to grant a license.
- While deciding on the application, the Controller will consider:
(i) Nature of the invention, time since the patent was granted, and steps taken by the patent holder to use the invention.
(ii) If the applicant can use the invention in a way that helps the public.
(iii) If the applicant has the resources and is ready to take the financial risk.
(iv) Whether the applicant tried to get a licence from the patent owner on fair terms and failed within a reasonable time (usually not more than 6 months).
However, in cases of national emergency, urgent need, public use, or if the patentee has used anti-competitive practices, this condition does not apply.
- The public’s needs are considered not met if:
(a) The patentee refuses fair licences, which:
-
- harms existing/new Indian businesses or industries,
- fails to meet demand,
- limits exports from India,
- harms commercial activities in India.
(b) The patent holder sets unfair conditions that hurt Indian trade or the use of non-patented materials.
(c) The patent holder forces:
-
- exclusive licence returns (grant-backs),
- bans on challenging the patent, [7][8]
- or bundled licences (coercive packages)
(d) The invention is not used enough in India or not as much as reasonably possible.
(e) The use of the invention in India is restricted due to imports by:
-
-
- the patentee,
- those buying from the patentee,
or others whom the patentee has not acted against[9] [10]
Section 88 of the Indian Patents Act,1970 (Powers of Controller in granting compulsory licences) [11] [12]
(1) If the patent owner has imposed unfair conditions, the Controller can allow the applicant and their customers to use the patent
(2) If the applicant had a license and applied again, the Controller can cancel the previous license or change it.
(3) If the patent owner has more than one patent and the public’s needs are not being met for some of the patents or they cannot be used without the use of others, the Controller can provide permission to use the other patents too.
(4) f the terms and conditions of a licence were set by the Controller, and the licensee has commercially used the invention for at least 12 months, and finds that:
- The conditions have turned out to be harsher or more difficult than expected,
- And this is causing a financial loss,
- Then the licensee may apply to the Controller to revise the licence terms.
It should be noted that the request can only be made once , no second application is allowed.
Section 92 of the Indian Patents Act,1970 (Compulsory licences in Emergencies)[13] [14]
The Central Government by notification in the Official Gazette can grant compulsory license at the time of national emergency, extreme emergency or at the time of public use. This can apply to one patent or a group of patents. After the Controller grants the patent , the applicant is free to use the patent. However, it should be noted that the product must be priced at the lowest possible cost and the patentee must get a reasonable return for their invention.
This helps establish a balance between public interest and the rights of the patent holder. [15]
Section 92 A of the Indian Patents Act,1970 (Compulsory licences for Export of Pharmaceuticals) [16] [17]
The provisions under Section 92A allows the Indian pharmaceutical companies to manufacture and export the medicines to other countries in case of emergencies.
Such exports are permitted in a case where receiving country is experiencing a global crisis, and the country lacks the capacity to produce the medicines themselves.
The importing country must demonstrate that they have obtained a compulsory license to import the drugs/medicines, or they are legally authorized to import under their country’s domestic laws.
This ensured that the countries have access to medicines during emergencies while still respecting the patent laws.[18]
Africa HIV-AIDS case and Doha declaration
The HIV/ AIDS epidemic in Africa created an urgent need to import the cheaper solutions through importing the medicines from other countries. However, this triggered several disputes with major multinational companies and raised concerns over the violations of TRIPS agreement. The intervention by the WHO helped the process to be completed smoothly. Similar circumstances observed in Brazil increased the awareness to the inadequacies in the TRIPS agreement. These developments led to the implementation of the Doha Declaration on the TRIPS Agreement and Public Health in November 2001[19].[20]
The Doha Declaration brought more awareness about the major inadequacies and shortcomings in the TRIPS agreement. The declaration stated that no country should be deprived of adequate care and TRIPS should strive to achieve it. At the same time it should be in compliance with the rules of WTO. The Doha Declaration affirmed the right of WTO member states to issue the compulsory licenses in situations of public health emergencies such as HIV/AIDS. Paragraph 6 of the Declaration[21] recognized that LDCs might struggle to utilize CL effectively due to limited manufacturing capability, prompting the 2003 WTO waiver.
The Declaration on TRIPS Agreement and public health was a key factor behind the introduction of Section 92 A[22] of the Indian Patent Act. The Section 92A was introduced in response to the decision of WTO on compulsory licensing for the export of patented pharmaceutical products to countries with insufficient or no manufacturing capacity.[23] [24]
The declaration also helped in the enforcement of a new waiver that led to the establishment of Article 31bis in 2017 [25]. The purpose of the article is to enable countries like India to export affordable generic medicines, primarily for the treatment of HIV/AIDS, to least developed countries.
The establishment of Section 92 A has helped underdeveloped countries such as Rwanda, Zimbabwe and Cameroon to acquire the affordable medicines for the treatment of HIV/AIDS. Indian pharmaceutical companies such as Cipla and Hetero that manufacture generic medicines have played a crucial role in global access of the medicines to antiretrovirals (ARVs). These efforts have drastically reduced the treatment costs from around $10,000 per patient per year in 2000 to under $150 by 2006, thus proving the effectiveness of Section 92A, along with the Doha declaration in supporting the global health rights.[26][27]
Section 94 of the Indian Patents Act,1970 (Temporary Compulsory License)[28] [29]
The compulsory license may be revoked by an interested party or the patent holder by filling out the Form 21 and submitting it along with the evidence of the original conditions under which the compulsory license was granted and the changed conditions requiring the cancellation of the license. Additionally, a copy of the application and evidence has to be submitted to the individual holding the compulsory licence and the Controller must be informed after the service is completed.
The individual holding the license has a right to file an objection within one month from the date when the Controller receives the application for cancellation.
Finally, the Controller may choose to schedule a hearing before making a final decision. In case the Controller decides to terminate the compulsory licence, both the parties will recieve a formal order with any terms and conditions related to the termination.[30]
Case Laws
Bayer Corporation v. Union of India & Others (IPAB, 2013)
Facts of the Case –
Bayer Corporation held an Indian patent for Sorafenib a drug used to treat liver and kidney cancer (marketed as Nexavar), where Bayer had fixed the price of Nexavar at ₹2.8 lakh/month, making it unaffordable to majority of the patients in India.
Thus, Nacto Pharma, an Indian pharmaceutical company that manufactures generic medicines applied for a compulsory license to manufacture and sell the drug at ₹8,880/month, under the provision of Section 84 of the Indian Patents Act,1970 [32]
It is to be noted that Bayer Corporation had refused the request of Natco Pharma for a voluntary license.[33]
Controller’s Decision-
The Controller of Patents had granted the first compulsory license in India to Nacto Pharma.
The decision held that Bayer Corporation failed to:
-
- Meet the reasonable public requirement.
- Provide the drug at a reasonably affordable price.
- Work the patent in India (i.e., no local manufacturing).
Thus, the Controller ordered Natco Pharma to pay 6% royalty on sales to Bayer Corporation.
Issues on Appeal (by Bayer)-
Fulfilment of procedural requirements by Natco Pharma under Section 84.
- Whether Bayer’s pricing and distribution were sufficient.
- Whether infringement by another company (CIPLA) could be considered in meeting public demand.
- Whether the royalty was too low.
IPAB Ruling (March 2013)-
The IPAB upheld the order of the Controller to grant the compulsory license and increased the royalty amount to 7%.
It was upheld that “Reasonable affordability” must be assessed from the perspective of the patients from India. CIPLA was not required to meet the public requirement that must be met by the patentee.
- The working requirement means actual availability, preferably through local manufacture.
- A single refusal of a voluntary license request is sufficient.
It was held that public interest and access to essential medicine takes precedence over the monopoly rights of the patentee.[34] [35]
Lee Pharma v. AstraZeneca (C.L.A. No. 1 of 2015)
Facts of the Case-
- Lee Pharma, an Indian pharmaceutical company manufacturing generic medicines, applied for a compulsory license (CL) to manufacture and sell SAXAGLIPTIN, a drug for Type-II Diabetes, sold by AstraZeneca under brand names Onglyza and Kombiglyze XR. The patent for SAXAGLIPTIN was previously granted to Bristol Myers Squibb (BMS), and was later assigned to AstraZeneca in 2014.
- Lee Pharma filed the CL application under the provision of Section 84(1) citing:
-
- Reasonable public requirements are unmet.
- Unaffordability of the drug.
Further, the patent was not being worked in India.
Controller’s Findings-
The Controller held that similar diabetes medications were already available in the market, and therefore the argument of Lee Pharma for the grant of compulsory license was not upheld. It was also found that both companies priced their products in the same range. AstraZeneca was importing the medicine into India, which was deemed sufficient to indicate as “working” the patent, whereas Lee Pharma failed to provide sufficient evidence to show otherwise.
Outcome-
The Controller rejected the compulsory license application.
- Held that no prima facie case was made under Section 84(1).
- Decision favoured AstraZeneca, stating the drug SAXAGLIPTIN was not eligible for compulsory licensing.[3
India and TRIPS agreement and its effect on compulsory licensing
Before joining the TRIPS agreement[37] [38] India would allow new drugs to be sold without patent protection. After joining TRIPS agreement, great protection was given to the patent right holder in the aspects of quality, availability and pricing of the drugs.
India had also added a feature of comprehensive compulsory license to protect public health and ensure availability of affordable medicines.[39]
COMPULSORY LICENSE OF PATENTS IN EU
Introduction
The EU has made a proposal wherein the European Commission can grant compulsory license, and additional protections would be provided during an emergency or a crisis, thus increasing the availability of the medicines to individuals during a crisis.
The European Parliament has stated that initially the companies will be given an option to grant a compulsory license voluntarily. They will have a period of 4 weeks to decide before a compulsory license is issued. This regulation excludes gas, chips and defence. It should also be noted that the licensee will bear the responsibility for any damages. The licensee will be liable for an amount between €300,000 (or €50,000 for small businesses) .[40]
The Commission also has two major roles. Firstly, it must explain itself if it does not follow expert advice. Secondly, it must identify the patent owner.
Patent owners must also be duly paid and duly on time. A 4% cap on payments to patent owners has been removed, except in special cases such as medicine exports in health emergencies.
The Commissioner may ask the patent owner to share relevant information, where the patent owner has a provision for receiving a compensation for the use of their patent.[41]
Compulsory Licensing for Pharmaceuticals
During the rise of the COVID-19 pandemic many questions arose about the compulsory licensing of patents. It was contended that licensing should be provided during emergencies.
Even though EU provided compulsory licensing many challenges still preside –
- There is no EU wide license available – Each EU country makes its own laws regarding compulsory licensing and that law is applicable only in that specific country. The EU itself cannot provide licenses which are applicable to every Member State.
- Article 31bis of TRIPS[42] [43] allows countries to import drugs that are under the preview of compulsory licenses. Unfortunately, EU had decided not to follow the TRIPS regulations, therefore an EU country is now allowed to import drugs from other countries. Further, EU is not allowed to import drugs from other EU member states. Therefore, a country under European Union (EU) solely relies on its own domestic production. [44]
- In order to sell a medicine, the companies must provide data to the European Medicines Agency (EMA). The data is protected for a period of 8 years and is protected from the public unless a waiver affiliated with it. Without a waiver, even if a licence is granted, the licensee is prohibited from accessing the data required to gain approval.
- In Belgium, the private entities are allowed to seek a compulsory license whereas the government is prohibited to do so. This regulation is heavily criticised stating that it hinders the process during the times of emergencies relating to public health.
An alternative to this could be the voluntary granting of the compulsory licenses by the patent holders or groups such as the WHO COVID-19 patent pool and Open COVID Pledge[45] encouraging the companies to share their patents freely for the greater good. Such provision ensures a faster and more cooperative way to access informaiton across borders.
The EU has legal tools for compulsory licensing, but they are complicated and limited. A better coordination, updated rules and regulations, and the possibility of rejoining the TRIPS import system could offer support. But for now, the voluntary solutions such as patent pools remains the most practical way forward.[46]
Case Law
Merck Sharp and Dohme v. Shionogi (2017, German Federal Court of Justice)
A US based pharmaceutical company, Merck Sharp and Dohme or MSD was supplying an HIV drug called Isentress (containing Raltegravir) in Germany. A Japanese company, Shionogi held a European patent for Raltegravir in Germany. Shionogi informed MSD regarding the patent infringement caused by Isentress, where MSD eventually proposed a one-time global license payment of $10 million USD, which Shionogi rejected as insufficient. The negotiations lasted a period of 2 years, without reaching an agreement.
In August 2015, Shionogi filed a patent infringement suit in the District Court of Düsseldorf. MSD responded by initiating opposition proceedings to invalidate the patent. In 2016, MSD sought a compulsory license under Section 24 of the German Patent Act[48][49], and a preliminary injunction under Section 85 PatG[50] [51] to continue selling Isentress.
On 31 August 2016, the Federal Patent Court granted MSD a preliminary compulsory license and this decision was upheld by the Federal Court of Justice on 11 July 2017.[52] [53]
COMPULSORY LICENSE OF PATENTS IN US
Introduction
Many countries have opted for compulsory licensing due to their being an inadequacy of supply, public interest and use within a country.
However, the US has not completely embraced this approach. Instead it relies on a similar version called the March-in Rights, established under the Bayh-Dole Act, 1980[54][55]. The law states that ‘if an invention is made in the US, the government has the authority to“march in” and allow another party to use the invention provided that the invention is not made in their country and the invention has not been made available to the public.
Important Provisions
- 28 U.S.C. § 1498(a)[56][57] – Government Use Provision: The statute allows the government to access any patented invention without authorization, provided the patent holder is compensated by a reasonable compensation. Although not labelled as compulsory licensing, this provision overrides the exclusive patent rights in favour of public interest or federal need.
- 35 U.S.C. § 203 – March-in Rights (Bayh-Dole Act, 1980)[58] [59] – This provision is deemed to be the closest formal mechanism to compulsory licensing. It grants the government the right to license out federally funded inventions if the patent holder has failed to achieve practical applications, public health/safety needs, public use requirements, or grants an exclusive license to an entity that does not manufacture substantially in the U.S., or the licensee breaches that obligation.[60]
Case Laws
University of Rochester v. G.D. Searle & Co. case (69 U.S.P.Q.2d 1996, Fed. Cir. 2004)
In this case the University of Rochester had patented a method to block an enzyme that was causing inflammation (COX-2). A company, Pfizer was selling COX-2 inhibitors such as Celebrex and Bextra, which are the drugs that operates on the same mechanism described in the patent of the University of Rochester. [62]
The University obtained the permission to patent and subsequently filed a lawsuit on Pfizer for patent infringement. They contended that Pfizer was using their method without permission and therefore demanded compensation and an injunction.
Pfizer argued that the University did not include any information about a specific drug or invention in their application instead only the method was mentioned.
It was finally decided that Pfizer did not embark in patent infringement as the University failed to follow the proper patenting procedures. [63][64]
Recent Developments
COMPULSORY LICENSE OF PATENTS IN JAPAN
A compulsory license in Japan is a legal mechanism under the Japanese Patent Act that allows a third party to use a patented invention without the consent of the patent holder under specific conditions, balancing patent rights with public interest.
Legal Provisions
Article 83[69] – Non-Working Patents: If the patent has been filed in a span of 3-4 years and is not “worked” (i.e., put to use), any individual has the authority to request a non-exclusive compulsory license after attempting negotiations.
Article 92[70] – Utilization Relationship Between Patents: If a patent holder cannot practice their invention without infringing the rights of others, both the parties may request mutual compulsory licenses, if a mutual agreement is not reached after the negotiations.
Article 93[71] – Public Interest: If a good-faith negotiation fails, an individual can seek permission for a compulsory license for public interest benefits.[72]
Case Laws
Landmark Case: Vision Care Inc. and VC Cell Therapy Inc. (2021)
In this case, Patent No.6518878 covering a method to produce RPE cells from iPS cells was co-owned by Helios Inc., RIKEN, and Osaka University and was exclusively licensed to Sumitomo Pharma.
Dr. Masayo Takahashi, the inventor, left RIKEN to lead Vision Care Inc. (VCCT) to accelerate the implementation. The negotiations between VCCT and Helios failed to reach a mutual agreement. Therefore, a compulsory license was requested under Article 93(2) of the Japanese patent law, and the application also focused on the aspect of public interest. The Japan Patent Office (JPO) provisionally supported the request but encouraged voluntary discussions.
It was also stated that Sumitomo had failed to follow the required process. Finally, a decision was reached wherein VCCT would gain the right to use the patent for non-insurance-based treatments with autologous iPS cells, and the patent owners agreed not to enforce certain rights under specified conditions.
Though no formal license was granted, the threat of one helped secure access.[74]
CONCLUSION
To conclude, each country aims to strike a balance between public interest and patent rights. India and Japan have poignant and well-defined rules on compulsory licensing with a strong focus on medicines, whereas the EU faces legal fragmentation and the US relies on alternative tools. Stronger global cooperation and alignment are required to ensure equal access of the information.
Author: Ishika Sarah Koshy , REVA University, Bangalore
Please contact us at info@origiin.com to know more about our services (Patent, Trademark, Copyright, Contract, IP Licensing, M&A of companies)
Subscribe to YouTube Channel HERE
Join LinkedIn Group: Innovation & IPR
WhatsApp: +91 74838 06607
 The growing global demand for clean and sustainable energy has driven significant interest in harnessing the power of the oceans. Sea energy, encompassing technologies such as wave energy, tidal energy, ocean thermal energy conversion (OTEC), and salinity gradient power, represents a promising frontier in renewable energy innovation. As countries and corporations invest in marine energy solutions, the patent landscape has become a critical indicator of technological advancements, competitive positioning, and commercialization potential.
The growing global demand for clean and sustainable energy has driven significant interest in harnessing the power of the oceans. Sea energy, encompassing technologies such as wave energy, tidal energy, ocean thermal energy conversion (OTEC), and salinity gradient power, represents a promising frontier in renewable energy innovation. As countries and corporations invest in marine energy solutions, the patent landscape has become a critical indicator of technological advancements, competitive positioning, and commercialization potential.






















 The application of
The application of  Figure 1 : Patent filing trend between the years 2014 to 2024.
Figure 1 : Patent filing trend between the years 2014 to 2024.








 Previously, applications would be validated and maintained through separate patents, meaning that each member state had to grant patent following the provisions established under the
Previously, applications would be validated and maintained through separate patents, meaning that each member state had to grant patent following the provisions established under the  The legal framework for compulsory licensing are established through international agreements such as
The legal framework for compulsory licensing are established through international agreements such as 

