 In this case, Sun Pharma Laboratories (hereinafter referred to as ‘Sun Pharma’), one of the leading Indian pharmaceutical company, came before court for the relief of permanent injunction to restrain BDR Pharmaceuticals (hereinafter referred to as ‘BDR’) from using trademark which is structurally and phonetically similar to that of Sun Pharma’s mark. Brief facts of the case are as follows:
In this case, Sun Pharma Laboratories (hereinafter referred to as ‘Sun Pharma’), one of the leading Indian pharmaceutical company, came before court for the relief of permanent injunction to restrain BDR Pharmaceuticals (hereinafter referred to as ‘BDR’) from using trademark which is structurally and phonetically similar to that of Sun Pharma’s mark. Brief facts of the case are as follows:
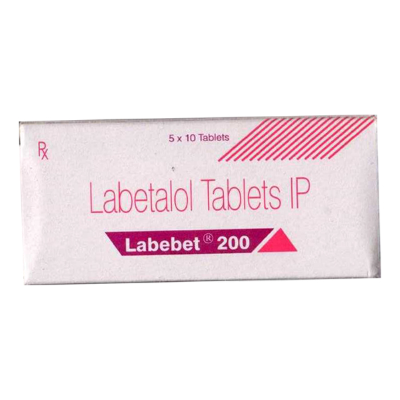
- Sun Pharma is carrying business of manufacturing and marketing pharmaceutical products for several decades and in the year 2009 they came out with the product LABEBET. LABEBET contains LABETALOL salt which is primarily used for treating hypertension and related issues. They also got the mark “LABEBET” registered in class 5 on 30th October, 2009 and since then they have been using this mark to sell their product in the market.
- Sun Pharma while performing trademark search in the registry came across application of the mark “LULIBET” dated 30th May, 2016, which was used by the BDR Pharmaceutical to market their product. And thus, Sun Pharma brought a suit of injunction to restraint BDR from further uses the mark “LULIBET” which is deceptively similar to registered mark of Sun Pharma “LABEBET”.
Arguments by the parties
By Plaintiff i.e., Sun Pharma
- That Sun Pharma is registered proprietor of the trademark “LABEBET” in class 5 since 2009 for medical and pharmaceutical preparation and is entitled to exclusive right of using the same by the virtue of section 28 of the Trade Marks Act, 1999 and that the action of BDR by using deceptively similar mark amounts to infringement by the virtue Section 29 of the Trade Marks Act.
- That the mark “LABEBET” when compared with the mark of BDR i.e., “LULIBET”, is structurally and phonetically similar.
- That both the products are sold in retail chemist shop, hence confusion and deception are likely to occur. Further, the possibility of patients with fungal infection consuming tablet of Sun Pharma cannot be ruled out. Thus, applying the test of imperfect recollection there is no doubt that public at large are bound to get deceived with such similarities.
- That Sun Pharma could not oppose the application of BDR when it was filed before Trade Mark Registry as the publication inadvertently escaped their attention, hence the present suit.
- That the contentions of BDR, that the marks are based on the salt/molecule from which product is formed, and that there are various other similar products available closely resembling to the mark of Sun Pharma against whom no action was initiated does not strengthen their arguments.
By Defendants i.e., BDR
- That BDR is the honest user of the mark, their mark is not deceptively similar to the mark of Sun Pharma, and that their mark was published in the Trademark Journal in the year 2016 which was not opposed by Sun Pharma and they cannot seek injunction for the same at the later stage.
- That the BDR derived the mark “LULIBET” from the salt/molecule upon which the product is based i.e., ‘Luliconzole’ which is used as lotion/cream for fungal treatment whereas Sun Pharma’s mark is based upon the molecule ‘Labetatol’ which is used as tablet/injection for curing hypertension. This, according to BDR form a crucial factor while comparing both the marks.
- That Sun Pharma did not coin the mark LABEBET, it is just based upon the salt/molecule from which product is derived and that there are large number of companies having trademark with the prefix LABE.
- That till the date of suit, no confusion was reported for the said marks and further the subsequent delay on part of Sun Pharma to initiate litigation shows that they are only attempting to stop progress of BDR Pharmaceuticals.
Issues
The main issues before Hon’ble Delhi High Court were:
- Whether the mark of BDR is structurally and phonetically with the mark of Sun Pharma?
- Whether the delay caused in filing opposition to the mark of BDR will deter Sun Pharma from claiming injunction?
Judgement
Court relying on various judgements held that:
“The marks have to be compared as a whole. They have to be judged by their look and their sound. The nature of customers who are likely to buy the goods has also to be considered in my opinion. If the two marks are compared as a whole the mark of the defendant is phonetically, visually and structurally, similar to that of the plaintiff. A person of average intelligence and imperfect recollection is likely to be deceived or confused. That apart as noted by the Supreme Court in Cadila Health Care Limited vs. Cadila Pharmaceuticals Ltd. (2001(5) SCC 73) where the medicinal products are involved the test to be applied would be stricter than should be applied for non-medicinal products. In the case of non-medicinal products, a confusion only creates economic loss but in the case of medicinal products, it may have adverse consequences on the health and life of the individual”.
It was further observed that delay in objecting or delay in bringing an action would not be sufficient to deter Sun Pharma from claiming injunction. And as such medical products are involved in the case which are sold through retail medical shops, court is bound to adopt stricter approach and thus the claim of BDR that both the product is sold in different form and has different usage will not make them escape liability.
No solid grounds were ultimately found to refuse injunction and thus decree was made in favour of Sun Pharma.
By: Dhruv Dangayach, Ramaiah College of Law
Please contact us at info@origiin.com to know more about our services (Patent, Trademark, Copyright, Contract, IP Licensing, M&A of companies)
Subscribe to YouTube Channel HERE
Join Linkedin Group: Innovation & IPR
Whatsapp: +91 74838 06607